2.3.2 Tubular Flow Reactor (PFR)
We model the tubular reactor as having the fluid flowing in plug flow—i.e., no radial gradients in concentration, temperature, or reaction rate.1 As the reactants enter and flow axially down the reactor, they are consumed and the conversion increases along the length of the reactor. To develop the PFR design equation, we first multiply both sides of the tubular reactor design equation (1-12) by –1. We then express the mole balance equation for species A in the reaction as
![]()
For a flow system, FA has previously been given in terms of the entering molar flow rate FA0 and the conversion X
![]()
Differentiating
dFA = –FA0dX
and substituting into (2-14) gives the differential form of the design equation for a plug-flow reactor (PFR):
![]()
We now separate the variables and integrate with the limits V = 0 when X = 0 to obtain the plug-flow reactor volume necessary to achieve a specified conversion X:
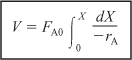

To carry out the integrations in the batch and plug-flow reactor design equations (2-9) and (2-16), as well as to evaluate the CSTR design equation (2-13), we need to know how the reaction rate –rA varies with the concentration (hence conversion) of the reacting species. This relationship between reaction rate and concentration is developed in Chapter 3.
