8.5.1 Complex Reactions in a PBR
Example 8-5. Multiple Gas Phase Reactions in a PBR
The following complex gas phase reactions follow elementary rate laws
![]()
and take place isothermally in a PBR. The feed is equimolar in A and B with FA0 = 10 mol/min and the volumetric flow rate is 100 dm3/min. The catalyst weight is 1,000 kg, the pressure drop is α = 0.0019 kg–1, and the total entering concentration is CT0 = 0.2 mol/dm3.
![]()
a. Plot and analyze FA, FB, FC, FD, y, and ![]() as a function of catalyst weight, W.
as a function of catalyst weight, W.
Gas Phase PBR
Mole Balances
![]()
![]()
![]()
![]()
Net Rates
![]()
![]()
![]()
![]()
Rate Laws
![]()
![]()
Relative Rates
Reaction 1: ![]()
![]()
![]()
Reaction 2: ![]()
![]()
![]()
The net rates of reaction for species A, B, C and D are


Selectivity
![]()
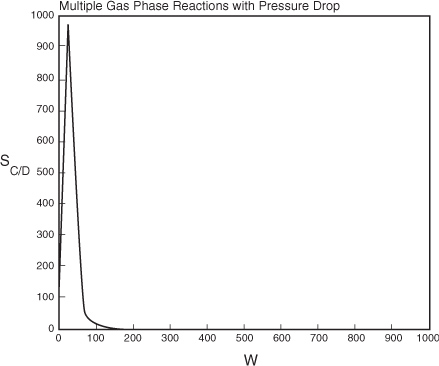
At W = 0, FD = 0 causing SC/D to go to infinity. Therefore, we set SC/D = 0 between W = 0 and a very small number, W = 0.0001 kg to prevent the ODE solver from crashing. In Polymath this condition is written
![]()
Stoichiometry Isothermal T = T0
![]()
![]()
![]()
![]()
![]()
![]()
Parameters
(22) CT0 = 0.2 mol/dm3
(23) α = 0.0019 kg–1
(24) υ0 = 100 dm3/min
(25) k1A = 100(dm3/mol)2/min/kgcat
(26) k2C = 1,500(dm15/mol4)/min/kgcat
(27) FT0 = 20 mol/min
Typing the above equations into Polymath’s ODE solver, we obtain the following results in Table E8-5.1 and Figures E8-5.1 and E8-5.2.
Analysis: We note from Figure E8-5.2 that the selectivity reaches a maximum very close to the entrance (W ≈ 60 kg) and then drops rapidly. However, 90% of A is not consumed until 200 kg, the catalyst weight at which the desired product C reaches its maximum flow rate. If the activation energy for reaction (1) is greater than that for reaction (2), try increasing the temperature to increase the molar flow rate of C and selectivity. However if that does not help, then one has to decide which is more important, selectivity or the molar flow rate of the desired product. In the former case the PBR catalyst weight will be 60 kg. In the latter case the PBR catalyst weight will be 200 kg.
Table E8-5.1. Polymath Program and Output

![]()
PBR Results
Figure E8-5.1. Molar Flow Rate Profiles
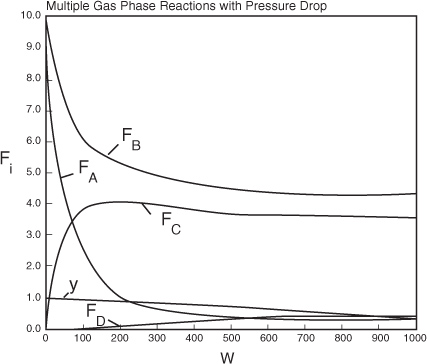
Figure E8-5.2. Selectivity Profile