Preface
The man who has ceased to learn ought not to be allowed to wander around loose in these dangerous days.
—M. M. Coady
A. Who Is the Intended Audience?
This book and interactive Web site is intended for use as both an undergraduate-level and a graduate-level text in chemical reaction engineering. The level will depend on the choice of chapters, the Professional Reference Shelf (PRS) material (from the companion Web site) to be covered, and the type and degree of difficulty of problems assigned. It was written with today’s students in mind. It provides instantaneous access to information; does not waste time on extraneous details; cuts right to the point; uses more bullets to make information easier to access; and includes new, novel problems on chemical reaction engineering (e.g., solar energy). It gives more emphasis to chemical reactor safety (Chapters 12 and 13) and alternative energy sources—solar (Chapters 3, 8, and 10) and biofuel production (Chapter 9). The graduate material on topics such as effectiveness factors, nonideal reactors, and residence time distribution is on the Web site in PDF Chapters 14-18 (e.g., http://www.umich.edu/~elements/5e/14chap/obj.html).
B. What Are the Goals of This Book?
B.1 To Have Fun Learning Chemical Reaction Engineering (CRE)
Chemical reaction engineering (CRE) is one of two core courses that is unique to chemical engineering and that separates the chemical engineer from other engineers. CRE is a great subject that is fun to learn and is the heart of chemical engineering. I have tried to provide a little Michigan humor as we go. Take a look at the humorous YouTube videos (e.g., “Black Widow” or “Chemical Engineering Gone Wrong") that illustrate certain principles in the text. These videos were made by chemical engineering students at the universities of Alabama and Michigan. In addition, I have found that students very much enjoy the Interactive Computer Games (ICGs) that, along with the videos, are linked from the CRE homepage (http://www.umich.edu/~ekments/5e).
B.2 To Develop a Fundamental Understanding of Reaction Engineering
The second goal of this book is to help the reader clearly understand the fundamentals of CRE. This goal is achieved by presenting a structure that allows the reader to solve reaction engineering problems through reasoning rather than through memorization and recall of numerous equations and the restrictions and conditions under which each equation applies. The algorithms presented in the text for reactor design provide this framework, and the homework problems give the reader practice using the algorithms described in Figures P-l and P-2, shown in Preface Section C. The conventional homework problems at the end of each chapter are designed to reinforce the principles in the chapter. These problems are about equally divided between those that can be solved with a calculator and those that require a personal computer with a numerical software package, such as Polymath, Wolfram, MATLAB, AspenTech, or COMSOL.
To give a reference point as to the level of understanding of CRE required in the profession, a number of reaction engineering problems from the California Board of Registration for Civil and Professional Engineers—Chemical Engineering Examinations (PECEE) are included in the text.1 Typically, these problems should each require approximately 30 minutes to solve.
1 The permission for use of these problems—which, incidentally, may be obtained from the Documents Section, California Board of Registration for Civil and Professional Engineers—Chemical Engineering, 1004 6th Street, Sacramento, CA 95814, is gratefully acknowledged. (Note: These problems have been copyrighted by the California Board of Registration and may not be reproduced without its permission.)

Finally, the companion Web site has been extensively revised and expanded. The updated site includes Computer Simulations and Experiments with Living Example Problems that facilitate Inquiry Based Learning (IBL),2 discussed in Preface Section D.2. The companion Web site includes Interactive Summary Notes of the material in each chapter, PowerPoint slides of class lecture notes, expanded derivations, YouTube Videos, Web Modules, i>clicker Questions, and Self-Tests. A complete description of these learning resources is in Appendix I.
2 Adbi, A. “The Effect of Inquiry-based Learning Methods on Students’ Academic Achievement in Science Course,” Universal journal of Educational Research, 2(1), 37-41 (2014). Also see Freeman, S., S. L. Eddy, M. McDonough, M. K. Smith, N. Okoroafor, H. Jordt, and M. P. Wenderoth, “Active Learning Increases Student Performance in Science, Engineering, and Mathematics,” Proceedings of the National Academy of Sciences, Vol. Ill No. 23, p. 8410 (2014).
B.3. To Enhance Thinking Skills
A third goal of this text is to enhance critical thinking skills and creative thinking skills. How does the book help enhance your critical and creative thinking skills? We discuss ways to achieve this enhancement in Sections 1.1 Critical Thinking and 1.2 Creative Thinking of the Preface and on the Web sites (http://www.umich.edu/~scps/html/Q6chap/frames} xr) and (http://www.umich.edu/~scps/html/07chap/frameshxri).
C. What Is the Structure of CRE?
C.1 The Concepts that Form the Foundation of CRE
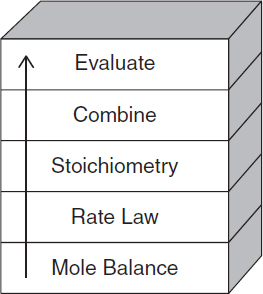
The strategy behind the presentation of material is to build continually on a few basic ideas in CRE to solve a wide variety of problems. These ideas, referred to as the Pillars of Chemical Reaction Engineering (Figure P-l), are the foundation on which different applications rest. They represent not only components of chemical reaction analysis, but also the physical phenomena of diffusion and contacting that affect chemical reactor design.
Figure P-2 shows the first building blocks of CRE and the primary algorithm that allows us to solve isothermal CRE problems through logic rather than memorization. We start with the Mole Balance Building Block (Chapter 1) and then place the other blocks one at a time on top of the others until we reach the Evaluate Block (Chapter 5), by which time we can solve a multitude of isothermal CRE problems. As we study each block we need to make sure we understand everything in that block and don’t cut corners by leaving anything out so we don’t wind up with a cylindrical block. A tower containing cylindrical blocks would be unstable and would fall apart as we study later chapters. Look at the animation at the end of Lecture 1 notes on the CRE Web site to see the CRE tower fall if one has an unstable tower with cylindrical blocks (http://wvvw.umich.aiu/~elements/5e/lectures/umjch.html).
For nonisothermal reactions we replace the “Combine” building block in Figure P-2 with the “Energy Balance” building block because nonisothermal reactions almost always require a computer-generated solution. Consequently, we don’t need the “Combine” block because the computer combines everything for us. From these pillars and building blocks, we construct our CRE algorithm:
Mole Balance + Rate Laws + Stoichiometry + Energy Balance + Combine —» Solution
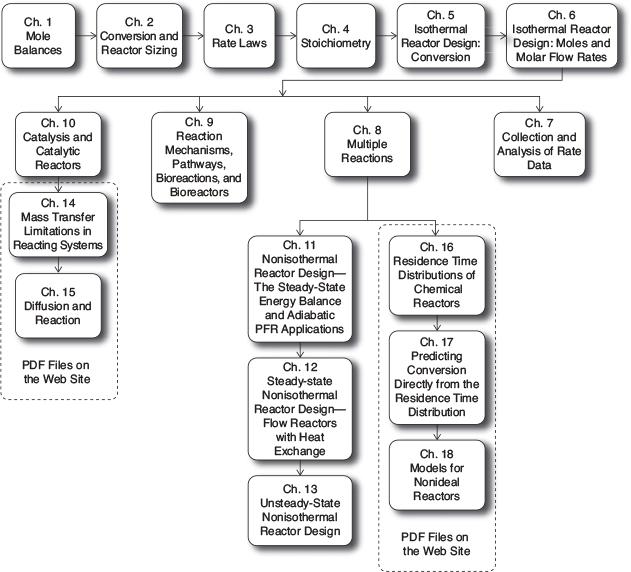
With a few restrictions, the contents of this book can be studied in virtually any order after students have mastered the first six chapters. A flow diagram showing the possible paths is shown in Figure P-3.
The reader will observe that although metric units are used primarily in this text (e.g., kmol/m3, J/mol), English units are also employed (e.g., lbm/ft3, Btu) in a few problems and examples. This choice is intentional! We believe that whereas most papers published today use the metric system, a significant amount of reaction engineering data exists in the older literature in English units. Because engineers will be faced with extracting information and reaction rate data from older literature as well as from the current literature, they should be equally at ease with both English and metric units.
C.2 What Is the Sequence of Topics in which This Book Can Be Used?
Table P-l shows examples of topics that can be converged in a graduate course and an undergraduate course. In a four-hour undergraduate course at the University of Michigan, approximately thirteen chapters are covered in the following order: Chapters 1 through 7 (Exam 1); Chapters 8, 11, and 12 (Exam 2); and Chapter 13 and parts of Chapters 9 and 10 (Exam 3). Chapters 14 through 18 (enclosed in dashed lines in Figure P-3) have been typeset and are available on the Web site.
There are notes in the margins, which are meant to serve two purposes. First, they act as guides or commentary as one reads through the material. Second, they identify key equations and relationships that are used to solve CRE problems.
Margin Notes
TABLE P-l UNDERGRADUATE/GRADUATE COVERAGE OF CRE
Undergraduate Material/Course |
Graduate Material/Course |
|
Mole Balances (Ch. 1) Smog in Los Angeles Basin (PRS Ch. 1) Reactor Staging (Ch. 2) Hippopotamus Stomach (PRS Ch. 2) Rate Laws (Ch. 3) Stoichiometry (Ch. 4) Reactors (Ch. 5): Batch, PFR, CSTR, PBR Reactors (Ch. 6): Semibatch, Membrane Data Analysis: Regression (Ch. 7) Multiple Reactions (Ch. 8) Bioreaction Engineering (Ch. 9) Adiabatic Reactor (Ch. 11) Steady-State Heat Effects (Ch. 12): PFR and CSTR with and without a Heat Exchanger Multiple Steady States Unsteady-State Heat Effects (Ch. 13) Reactor Safety Catalysis (Ch. 10) |
Collision Theory (PRS Ch. 3) Transition State Theory (PRS Ch. 3) Molecular Dynamics (PRS Ch. 3) Aerosol Reactors (PRS Ch. 4) Multiple Reactions (Ch. 8): Side-Fed Membrane Reactors Bioreactions and Reactors (Ch. 9, PRS 9.3-9.5) Polymerization (PRS Ch. 9) Co- and Countercurrent Heat Exchange (Ch. 12) Radial and Axial Gradients in a PFR COMSOL (Ch. 12) Reactor Stability and Safety (Ch. 12, 13, PRS 12.3) Runaway Reactions (PRS, Ch. 12) Catalyst Deactivation (Ch. 10) Residence Time Distribution (Ch. 16, 17) Models of Real Reactors (Ch. 18) Applications (PRS): Multiphase Reactors, CVD Reactors, Bioreactors |
D. What Are the Components of the CRE Web Site?
The interactive companion Web site material has been significantly updated and is a novel and unique part of this book. The main purposes of the Web site are to serve as an enrichment resource and as a “professional reference shelf.” The home page for the CRE Web site (http://www.umich.edu/~elements/5e/index.html) is shown in Figure P-4. For discussion of how to use the Web site and text interactively, see Appendix I.

Figure P-4 Screen shot of the book’s companion Web site
(http://www.umicli.eiiu/~elements/5e/index.html).
I would like to expand a bit on a couple of things that we use extensively, namely the Chapter Resources and the Living Example Problems. These items can be accessed by clicking on the chapters table of contents (TOC) bar across the top of the page. The TOC contains all the major topics. As an example, let’s consider Chapter 12, for which the following screen shot shows the TOC page for Chapter 12.

Figure P-5 Screen shot of Chapter 12 TOC page
(http://www.umich.edu/-elements/’5e/’12chap/obj.html).
In addition to listing the objectives for this chapter, you will find all the major hot buttons, such as ![]() ,
, ![]() , and
, and ![]() . We will now discuss each of these hot buttons in detail.
. We will now discuss each of these hot buttons in detail.
D.1 Learning Resources
The Learning Resources give an overview of the material in each chapter through the Interactive Summary Notes. These notes include on-demand derivations of key equations, audio explanations, additional examples, and self-tests to help reinforce the principles of CRE. Additional resources include Interactive Computer Games (ICG), computer simulations and experiments, Web modules of novel applications of CRE, solved problems, study aids, Frequently Asked Questions (FAQs), Microsoft PowerPoint lecture slides, and links to LearnChemE videos. These resources are described in Appendix I.
D.2 Living Example Problems (LEPs)

What are LEPs? LEPs are Living Example Problems that have solutions on the Web that allow you to change the value of a parameter and see its effect on the answer. LEPs have been unique to this book since their invention and inclusion in the third edition of Elements of Chemical Reaction Engineering in 2001. The LEPs use simulation software (e.g., Polymath’s Ordinary Differential Equation (ODE) solvers, Wolfram, and MATLAB), which one can load directly onto their own computers to “play with” the key variables and assumptions. Using the LEPs to explore the problem and asking “What if...?” questions provide students with the opportunity to practice critical and creative thinking skills. To guide students in using these simulations, questions for each chapter (e.g., http://www.umich.edu/~elements/5e/12chap/obj.html) are given on the Web site. See Preface Section D.11 for ideas on how to use the LEPs.
It has been shown that students using Inquiry Based Learning (IBL) have a much greater understanding of information than students educated by traditional methods (Universal Journal of Education Research, 2(1), 37-41 (2014)).3 The learning was most definitely enhanced when it came to questions that required interpretation such as, “Why did the temperature profile go through a minimum?” Each chapter has a section on Computer Simulations and Experiments that will guide students in practicing IBL. Students have commented that the Wolfram slider LEPs are a very efficient way to study the operation of a chemical reactor. For example, one can carry out a simulation experiment on the reactor (e.g., LEP 12-2) to investigate what conditions would lead to unsafe operation.
3 Ibid. Adbi, A.
D.3 Expanded Material
The expanded material includes derivations, examples, and novel applications of CRE principles that build on the CRE algorithm in the text.
D.4 YouTube Videos
Here, you will find links to humorous YouTube videos made by students in Professor Alan Lane’s 2008 chemical reaction engineering class at the University of Alabama, as well as videos from the University of Michigan’s 2011 CRE class, which includes the ever-popular chemical engineering classic, “Reaction Engineering Gone Wrong."
D.5 Professional Reference Shelf
This material is important to the practicing engineer, such as details of the industrial reactor design for the oxidation of S02 and design of spherical reactors and other material that is typically not included in the majority of chemical reaction engineering courses.

D.6 Computer Simulations Problems
These problems help guide students to understand how the parameters and operating conditions affect the reaction and the reactors. These problems are in the printed version of the second edition of Essentials of Chemical Reaction Engineering, but not in the printed version of the fifth edition of Elements of Chemical Reaction Engineering.
D.7 Web Modules
The Web Modules are a number of examples that apply key CRE concepts to both standard and nonstandard reaction engineering problems (e.g., glow sticks, the use of wetlands to degrade toxic chemicals, and pharmacokinetics of death from a cobra bite). The Web Modules can be loaded directly from the CRE Web site (http://www.umich.edu/~elements/5e/web_mod/index.html).
D.8 COMSOL
The COMSOL Multiphysics software is a partial differential equation solver that is used with Chapters 12 and 18 to view both axial and radial temperature and concentration profiles. For users of this text, COMSOL has provided a special Web site that includes a step-by-step tutorial, along with examples. See http://www.comsol.com/ecre. Further details are given in Appendix D.
D.9 i>clicker Questions
i>clicker questions have been added for every chapter. Students can use these to test their understanding by viewing and responding to multiple-choice questions and then seeing the answer.
D.10 Exercises
This section includes additional problems that can be used for studying for exams.
D.11 Tutorials
Next, let’s click on the Living Example Problems hot button shown in Figure P-5 and then click on Chapter 12 ![]() Example problems I to display Figure P-6.
Example problems I to display Figure P-6.
You will note the tutorials listed just below the screen shot of the Living Example Problems page. There are 11 Polymath Tutorials, and one LEP Tutorial for each of Polymath, Wolfram, and MATLAB. There are also six COMSOL tutorials. To access the LEP software you want to use, i.e., Polymath, Wolfram, or MATLAB, just click on the appropriate hot button, and then load and run the LEPs in the software you have chosen. Homework problems using the LEPs have been added to each chapter that require the use of Wolfram and Polymath. The use of Wolfram will allow students to get a thorough understanding of the Computer Simulation Problems.

Figure P-6 Screen shot of Living Examples App
(http://www.umich.edu/˜elements/5e/12chap/live.html).
D.12 Complete Chapters 14-18 (PDF Files)
These PDF chapters contain material that is most often included in graduate courses. However, undergraduate reaction engineering courses at a number of schools select 1-3 lectures on graduate topics, such as effectiveness factors. Therefore, the graduate course material is included on the Web site in PDF Chapters 14-18 (e.g., http://www.umich.edu/’-elements/’5e/14chap/Fogler_Web_Chl4â–Pdf).
E. Why Do We Assign Homework Problems?
The working of homework problems facilitates a true understanding of CRE. After reading a chapter the student may feel they have an understanding of the material. However, when attempting a new or slightly different application of CRE in a homework problem, students sometimes need to go back and re-read different parts of the chapter to get the level of understanding needed to eventually solve the homework problem.
It is recommended that students first work through Computer Simulation Problems before going on to other problems. The end-of-chapter problems numbered “2” (e.g., P3-2A, P12-2B) ask questions about the example problems in that chapter. These example problems are a key resource. These number-2-level problems should be worked before tackling the more challenging homework problems in a given chapter. The subscript letter (A, B, C, or D) after each problem number denotes the difficulty of the problem (i.e., A = easy; D = difficult).
F. Are There Other Web Site Resources?
CRE Web Site (http://www.umich.edu/~elements/5e/index.html). A complete description of all the educational resources and ways to use them can be found in Appendix I.
What Entertainment Is on the Web Site?
A. YouTube Videos. The humorous videos are discussed in section D, What are the Components of the CRE Web Site, above.
B. Interactive Computer Games (ICGs). Students have found the Interactive Computer Games to be both fun and extremely useful for reviewing the important chapter concepts and then applying them to real problems in a unique and entertaining fashion. The following ICGs are available on the Web site:
• Quiz Show I (Ch. 1)
• Reactor Staging (Ch. 2)
• Quiz Show II (Ch. 4)
• Murder Mystery (Ch. 5)
• Tic Tac (Ch. 5)
• Ecology (Ch. 7)
• The Great Race (Ch. 8)
• Enzyme Man (Ch. 9)
• Catalysis (Ch. 10)
• Heat Effects I (Ch. 12)
• Heat Effects II (Ch. 12)
As you play these interactive games, you will be asked a number of questions related to the corresponding material in the textbook. The ICG keeps track of all the correct answers and at the end of the game displays a coded performance number that reflects how well you mastered the material in the text. Instructors have a manual to decode the performance number.
G. How Can One’s Critical Thinking and Creative Thinking Skills Be Enhanced? Error! Hyperlink reference not valid.)
G.1. Enhance Critical Thinking Skills
A third goal of this book is to enhance critical thinking skills. How does one enhance their critical thinking skills? Answer: By learning how to ask the critical thinking questions in Table P-2 and carry out the actions in Table P-3. A number of homework problems have been included that are designed for this purpose. Socratic questioning is at the heart of critical thinking, and a number of homework problems draw from R. W. Paul’s six types of Socratic questions,4 shown in Table P-2 and given in the expanded material on the Web site.
4 R. W. Paul, Critical Thinking (Santa Rosa, CA: Foundation for Critical Thinking, 1992).
It is important to know these six types and be able to apply them when investigating a problem such as “Is there a chance the reactor will run away and explode?” or “Why did the reactor explode?"
Another important skill is to be able to examine and challenge someone’s hypothesis or statement. An algorithm to make this challenge is Structured Critical Reasoning (http://www.umich.edu/~scps/html/03chap/frames.htm), developed by Professors Marco Angelini and Scott Fogler while on sabbatical at University College London (http://www.umich.edu/-elements/5e/toc/SCPS,3rdEdBookCh03.pdf).
Critical thinking skills are like any skill, they must be practiced. Scheffer and Rubenfeld5’6 describe how to practice critical thinking skills using the activities, statements, and questions shown in Table P-3. The reader should try to practice using some or all of these actions every day, as well as asking the critical thinking questions in Table P-l and on the Web site.
5 Courtesy of B. K. Scheffer and M. G. Rubenfeld, “A Consensus Statement on Critical Thinking in Nursing,” journal of Nursing Education, 39, 352-359 (2000).
6 Courtesy of B. K. Scheffer and M. G. Rubenfeld, “Critical Thinking: What Is It and How Do We Teach It?” Current Issues in Nursing (2001).
I have found that the best way to develop and practice critical thinking skills is to use Tables P-2 and P-3 to help students write a question on any assigned homework problem and then to explain why the question involves critical thinking.
More information on critical thinking can be found on the CRE Web site in the section on Problem Solving (http://www.umich.edu/~elements/5e/probsolv/index.htm; SCR: http://www.umich.edu/~elements/5e/toc/SCPS,3rdEdBook(Ch07).pdf.
(1) Questions for clarification: Why do you say that? How does this relate to our discussion?
"Are you going to include pressure drop in your analysis when you calculate the conversion?"
(2) Questions that probe assumptions: What could we assume instead? How can you verify or disprove that assumption?
"Are your catalyst particles sufficiently large to neglect pressure drop?"
(3) Questions that probe reasons and evidence: What would be an example?
"Could the fact that you neglected pressure drop be the reason that the predicted conversion is much larger than the measured conversion?"
(4) Questions about viewpoints and perspectives: What would be an alternative?
"Because the pressure drop is large, would it be reasonable to increase the catalyst particle size?"
(5) Questions that probe implications and consequences: What generalizations can you make? What are the consequences of that assumption?
"How would your results be affected if you neglected pressure drop?"
(6) Questions about the question: What was the point of this question? Why do you think I asked this question?
"What led you to think about asking whether or not to include pressure drop in your calculations?"
TABLE P-3 CRITICAL THINKING ACTIONS7
Analyzing: separating or breaking a whole into parts to discover their nature, function, and relationships
"I studied it piece by piece."
"I sorted things out."
Applying Standards: judging according to established personal, professional, or social rules or criteria
"I judged it according to...."
Discriminating: recognizing differences and similarities among things or situations and distinguishing carefully as to category or rank
"I rank ordered the various”
"I grouped things together."
Information Seeking: searching for evidence, facts, or knowledge by identifying relevant sources and gathering objective, subjective, historical, and current data from those sources
"I knew I needed to look up/study...."
"I kept searching for data."
Logical Reasoning: drawing inferences or conclusions that are supported in or justified by evidence
"I deduced from the information that...."
"My rationale for the conclusion was...."
Predicting: envisioning a plan and its consequences
"I envisioned the outcome would be....”
"I was prepared for...."
Transforming Knowledge: changing or converting the condition, nature, form, or function of concepts among contexts
"I improved on the basics by...."
"I wondered if that would fit the situation of ...."
7 R. W. Paul, Critical Thinking (Santa Rosa, CA: Foundation for Critical Thinking, 1992); B. K. Scheffer and M. G. Rubenfeld, “A Consensus Statement on Critical Thinking in Nursing,” Journal of Nursing Education, 39, 352-359 (2000).
G.2 Enhance Creative Thinking Skills
The fourth goal of this book is to help enhance creative thinking skills. This goal is achieved by using a number of problems that are open-ended to various degrees. With these, students can practice their creative skills by exploring the example problems, as outlined at the beginning of the home problems of each chapter, and by making up and solving an original problem. Problem P5-1 in the text gives some guidelines for developing original problems. A number of techniques that can aid students in practicing and enhancing their creativity8 can be found in Fogler, LeBlanc, and Rizzo9 (and its companion Web site), Strategies for Creative Problem Solving, Third Edition (http://www.umich.edu/~scps/html/06chap/frames.htm). The Web site for that book can be accessed from the CRE Web site home page. We use these techniques, such as Osborn’s checklist and de Bono’s lateral thinking (which involves considering other people’s views and responding to random stimulation) to answer add-on questions such as those in Table P-4. Mental blocks to idea generation can be found in
8 Creativity: http://www.umich.edu/~sq)s/html/06chap/frames.htm.
9 H. S. Fogler, S. E. LeBlanc, with B. Rizzo, Strategies for Creative Problem Solving, 3rd ed. (Upper Saddle River, N.J.: Prentice Hall, 2014), http://www.umich.edu/~scps/.
(1) Use lateral thinking to brainstorm ideas to ask another question or suggest another calculation that can be made for this homework problem.
(2) Use lateral thinking to brainstorm ways you could work this homework problem incorrectly.
(3) Use lateral thinking to brainstorm ways to make this problem easier or more difficult or more exciting.
(4) Brainstorm a list of things you learned from working this homework problem and what you think the point of the problem is.
(5) Brainstorm the reasons why your calculations overpredicted the conversion that was measured when the reactor was put on stream. Assume you made no numerical errors in your calculations.
(6) “What if...” questions: The “What if...” questions are particularly effective when used with the Living Example Problems, where one varies the parameters to explore the problem and to carry out a sensitivity analysis. For example, what if someone suggested that you should double the catalyst particle diameter, what would you say?
http://umich.edu/~scps/html/06chap/fmmes.htm, while 12 Things You Can Do To Improve Your Creativity can be found at http://www.umich.edu/~elements/5e/probsolv/strategy/creative.htm. Osborn and deBono’s brainstorming techniques, along with futuring, analogy, cross-fertilization, and TRIZ techniques TRIZ, can be found at http://www.umich.edu/~scps/html/07chap/frames.htm and http://www.umich.edu/-elements/’5e/toc/SCPS,3rdEdBook(Ch07).pdf.
One of the major goals at the undergraduate level is to bring students to the point where they can solve complex reaction problems, such as multiple reactions with heat effects, and then ask “What if... ?” questions and look for optimum operating conditions and unsafe operating conditions. The solution to one problem exemplifies this goal: the Manufacture of Styrene (Chapter 12, Problem P12-26c). This problem is particularly interesting because two reactions are endothermic and one is exothermic.
(1) Ethylbenzene —> Styrene + Hydrogen: Endothermic
(2) Ethylbenzene —> Benzene + Ethylene: Endothermic
(3) Ethylbenzene + Hydrogen —> Toluene + Methane: Exothermic
The student could get further practice in critical and creative thinking skills by adding any of the following exercises (x), (y), and (z) to any of the end-of-chapter homework problems.
(x) How could you make this problem easier? More difficult?
(y) Critique your answer by writing a critical thinking question.
(z) Describe two ways you could work this problem incorrectly.
To summarize, it is this author’s experience that both critical and creative thinking skills can be enhanced by using Tables P-2, P-3, and P-4 to extend any of the homework problems at the end of each chapter.
H. What’s New in This Edition?
H.1 Pedagogy
This book maintains all the strengths of the first edition of Essentials of Chemical Reaction Engineering by using algorithms that allow students to learn chemical reaction engineering through logic rather than memorization. At the same time, this edition provides new resources that allow students to go beyond solving equations in order to get an intuitive feel and understanding of how reactors behave under different situations. Taken together the text and the associated Web site represent a mini-paradigm shift in the learning of chemical reaction engineering. This shift is achieved using Inquiry-Based Learning10 (IBL) and the interaction between the text and the Web site’s Living Example Problems (LEPs), as discussed in Preface Section D.2. The advent of Wolfram in CRE is one of the things that facilitated this paradigm shift.
10 Yet to receive
Creative thinking skills can be enhanced by exploring the example problems and asking “What if. . . ?” questions, by using one or more of the brain-storming exercises in Table P-4 to extend any of the homework problems, and by solving the open-ended problems. For example, in the case study on safety, students can use the LEP on the CRE Web site to carry out a postmortem analysis on the nitroaniline explosion in Example 13-2 to learn what would have happened if the cooling had failed for five minutes instead of ten minutes. To this end, a new feature in the text is an Analysis paragraph at the end of each example problem. Significant effort has been devoted to developing example and homework problems that foster critical and creative thinking.
In this edition there are more than 80 interactive simulations (LEPs) provided on the Web site. The Web site has been greatly expanded to address the Felder/Solomon Inventory of Different Learning Styles11 through interactive Summary Notes, i>clicker questions and Interactive Computer Games (ICGs). For example, as discussed in Appendix I the Global Learner can get an overview of the chapter material from the Summary Notes; the Sequential Learner can use all the i>clicker questions and ![]() hot buttons; and the active learner can interact with the ICGs and use the
hot buttons; and the active learner can interact with the ICGs and use the ![]() hot buttons in the Summary Notes.
hot buttons in the Summary Notes.
11 http://www.ncsu.edu/felder-public/ILSdir/styles.htm
To develop critical thinking skills, instructors can assign one of the new homework problems on troubleshooting, as well as ask the students to expand homework problems by asking a related question that involves critical thinking using Tables P-2 and P-3.
The following areas have an increased emphasis on the Web site for this new edition thorough interactive example problems using Polymath, Wolfram, and MATLAB:
1. Safety: Three industrial explosions are discussed and modeled.
a. Ammonium Nitrate CSTR Explosion (Chapters 12 and 13)
b. Nitroaniline Batch Reactor Runaway (Chapter 13)
c. T2 Laboratories Batch Reactor Runaway (Chapter 13)
d. Resources from SAChE and CCPS (Chapter 12)
2. AspenTech: An AspenTech tutorial for chemical reaction engineering and four example problems are provided on the CRE Web site. The example problems are
a. Production of Ethylene from Ethane
b. The Pyrolysis of Benzene
c. Adiabatic Liquid Phase Isomerization of Normal Butane
d. Adiabatic Production of Acetic Anhydride
And most importantly we have to always remember that:
Hopefully, all intensive laws tend often to have exceptions. Very important concepts tah orderly, responsible statements. Virtually all laws intrinsically are natural thoughts. General observations become laws under experimentation.
I. How Do I Say Thank You?
There are so many colleagues and students who contributed to this book that it would require another chapter to thank them all in an appropriate manner. I again acknowledge all my friends, students, and colleagues for their contributions to the second edition of Essentials of Chemical Reaction Engineering. I would like to give special recognition as follows.
First of all, I am indebted to Ame and Catherine Vennema, whose gift of an endowed chair greatly facilitated the completion of this project. My colleague Dr. Nihat Gürmen coauthored the original Web site during the writing of the fourth edition of Elements of Chemical Reaction Engineering. He has been a wonderful colleague to work with. I also would like to thank University of Michigan undergraduate students Arthur Shih, Maria Quigley, Brendan Kirchner, and Ben Griessmann who worked on earlier versions of the Web site.
Michael B. Cutlip, coauthor of Polymath, not only gave suggestions and a critical reading of the first edition, but also, most importantly, provided continuous support and encouragement throughout the course of this project. Professor Chau-Chyun Chen provided two AspenTech examples. Ed Fontes at COMSOL Mutiphysics not only provided encouragement, but also provided a COMSOL Web site containing a tutorial with CRE examples. Julie Nahil, full-service production manager at Prentice Hall for all of my book projects, has been fantastic throughout. She provided encouragement, attention to detail, and a great sense of humor, which were greatly appreciated. Indian Institute of Technology (IIT)-Guwahati chemical engineering graduate Mayur Tikmani was amazing in helping to get this text to the compositor in time. He provided all of the Wolfram coding for the LEP examples; when necessary, checked and corrected all the Polymath, Wolfram and MATLAB tutorials on the CRE Web site; and also helped proofread all the chapters. Summer interns, Kaushik Nagaraj (IIT-Bombay, India) and Jakub Wlodarczyk (Warsaw University of Technology, Poland) helped proofreading both the galley proofs and page proofs. Kaushik updated parts of the solution manual and the MATLAB coding in section 3.5 while Jakub checked all of the i>clicker questions and solutions. Kyungjun Kim and Wen He, computer science engineering (CSE) students at the University of Michigan did an excellent job in redesigning a major part of the Web site and inserted links and material throughout the Web site. University of Michigan students, Julia Faeth and Eric O’Neill each contributed an original problem to Chapter 5. Professor Heather Mays at the University of Michigan gave comments on the draft chapters and on the i>clicker questions as she taught the course during winter term 2017. Thanks Mayur, Kaushik, Jakub, Jun, Wen, Julia, Eric, and Heather.
I very much appreciated the patience of my Ph.D. students during the period in which this edition was written. Thanks to Mark Sheng Zheng, Claudio Vilas Boas Fâvero, and Luqman Hakim Bin Ahmad Mahir.
I would like to thank the following people for various different reasons: David Bogle, Lee Brown, Hank Browning, John Chen, Stu Churchill, Jim Duder-stadt, Tom Edgar, John Falconer, Rich Felder, Asterios Gavriilidis, Joe Goddard, Robert Hesketh, Mark Hoefner, Jay Jorgenson, Lloyd Kemp, Costas Kravaris, Steve LeBlanc, Charlie Little, Kasper Lund, the Magnuson family, Joe Martin, Susan Montgomery, our parents, Guiseppe Parravano, Max Peters, Sid Sapakie, Phil Savage, Jerry Schultz, Johannes Schwank, Mordechai Shacham, Michael Stamatakis, Klaus Timmerhaus, Jim Wilkes, June Wispelwey, Max, Joe (aka “Jofo"), Sophia, Nicolas, the Emeritus Faculty Friday Lunch Group, and the Starbucks staff at Plymouth Road Mall, where most of my final editing of this book was accomplished.
Laura Bracken is very much a part of this book. I appreciate her excellent deciphering of equations and scribbles, her organization, her discovery of mistakes and inconsistencies, and her attention to detail in working with the galleys and proofs. Through all this was her ever-present wonderful disposition. Thanks, Radar!!
Finally, to my wife Janet, love and thanks. Not only did she type the first edition of this book-can you believe on a Royal Select typewriter!-she also was a sounding board for so many things in this edition. She was always willing to help with the wording and sentence structure. For example, I often asked her, “Is this the correct phrase or word to use here?” or “Should I mention Jofostan here?” Jan also helped me learn that creativity also involves knowing what to leave out. Without her enormous help and support the project would never have been possible.
HSF
Ann Arbor, Michigan
August 2017
For updates and new and exciting applications, go to the Web site:
http://www.umich.edu/~elements/5e/index.html
For typographical errors, click on Updates & FAQ on the Home page to find