Anti-inflammatory Dietary Ingredients, Medicinal Plants, and Herbs Exert Beneficial Health Effects in Aging
Kiran S. Panickar and Dennis E. Jewell, Hill’s Pet Nutrition Center, Topeka, KS, United States
Abstract
Nutrition plays an important role in attenuating some of the detrimental effects of aging. Aging in humans is associated with chronic and systemic low-grade inflammation as well as a decline in mobility, joint problems, weakened muscles and bones, reduced lean body mass, cancer, increased dermatological problems, decline in cognitive ability, reduced energy, decreased immune function, decreased renal function, and urinary incontinence. Strategies to reduce inappropriate chronic inflammation have included dietary modification and the use of herbal extracts in the diet or as supplements. Canines also have general aging-associated health conditions that are similar to those in humans. The underlying causes of detrimental health in aging are likely to be many, but each condition is also associated with an increase in circulating pro-inflammatory markers. An inflammatory state characterized by an increase in pro-inflammatory markers includes tumor necrosis factor α, interleukins 6 and 1 β (IL-6 and IL-1β), and C-reactive protein. All are believed to contribute to or worsen aging-related general declines in the biological mechanisms responsible for physical function. Natural botanicals have bioactive components that appear to have robust anti-inflammatory effects and may contribute to a reduction in inflammation when included in the diet. Review of all aging-associated health conditions is beyond the scope of this chapter, so we will focus on selected areas of health conditions, including immune function, renal function, dementia, and cognition in animal and human studies. We will also review the scientific data on the anti-inflammatory effects of dietary ingredients as well as the efficacy of bioactive molecules from botanicals.
Keywords
Polyphenols; inflammation; cytokines; immune; botanical; inflammaging
Introduction
One hallmark of aging is chronic low-grade inflammation (Brüünsgaard and Pedersen, 2003; Candore et al., 2010) that is accompanied by an increase in circulating pro-inflammatory cytokines (Roubenoff et al., 1998; Bruunsgaard, 2002, Michaud et al., 2013). A pro-inflammatory state contributes to various aging-associated dysfunctions at the cellular and molecular levels.
The immune system is a key component of inflammation and is body’s natural response to injury or infection. Immune function involves several cell types that regulate both innate and adaptive responses as well as interactions among them while responding to an antigen. Innate immune response is nonspecific and relatively immediate whereas adaptive immune response is antigen-specific and delayed. Inflammation is generally classified as acute or chronic. While many features of the acute inflammatory response may also manifest themselves in chronic inflammation, there are distinguishing features. For instance, acute inflammation, as its name indicates, is a quick response to an infection or injury and is often resolved quickly; chronic inflammation may develop in days and can be progressive. While neutrophils play an important role in acute inflammation, monocytes, macrophages, and lymphocytes play a major role in chronic inflammation. One form of early response to infection or injury is the acute phase response (APR). The APR is characterized by the production of plasma protein-derived proteins such as C-reactive protein (CRP) and serum amyloids A and P, and they complement proteins by the liver generally in response to cell-derived mediators such as prostaglandins, nitric oxide, leukotrienes, and cytokines. The goal of the early response includes (1) destroying or inhibiting the activity of foreign bodies in particular microbes, (2) exerting protective effects that may limit infection, (3) removing necrotic tissue, and (4) initiating cell repair. This response is distinct from a state of mild inflammation, which is associated with chronically elevated levels of inflammatory markers, including C-reactive protein as well as cytokines such as interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α).
Immunosenescence is the gradual decline of the immune system with age (Franceschi et al., 2000). Both innate and adaptive responses are dampened with aging, and there is also a decline in the diversity of the antigen repertoire and an accumulation of functionally impaired memory lymphocytes that can affect the ability to fight infection or recover from injury. While there is consensus that the immune responses decline with aging, the specific changes in the repertoire of specific cell types leading to a compromised immune system are not clear. For instance, there might be a decrease in the levels of cluster of differentiation (CD) cells CD27 and CD28 and the generation of immature B cells but an increase in CD244 and memory B cells (see Alam and Pawelec, 2012 for review). This signifies the complexity of the immune system in aging. CD244, for instance, is a cell surface receptor on natural killer (NK) cells, which might imply increased NK-mediated cytolytic activity. In addition, this receptor can also be expressed on nonlymphocytes such as eosinophils, mast cells, and dendritic cells. Whereas taking this one example in isolation would indicate an increased immune function in aging, a generalized dampened immune function in aging is likely the resultant composite function of the immune system as a whole. In canines, there does not appear to be clear consensus on the types of leukocytes that decline with aging although a general decline in immune function with aging is observed. Kearns et al. (1999) reported an age-associated decline in the proliferative capacity of lymphocytes in fox terriers and Labrador retrievers, indicating a reduced immune activity. Fleming et al. (2011) reported that older dogs died of neoplastic, traumatic, and infectious disorders, which indicates a possibly reduced immune function and ability to fight infection. Nevertheless, in older canines an ability to mount a primary humoral response to novel antigens is generally retained but the magnitude of the response is likely reduced relative to titers achieved in younger animals (Day, 2010). Aged beagle dogs have decreased neutrophil phagocytosis when compared to young ones as assessed by their ability to phagocytize Lactococcus lactis ex vivo (Hall et al., 2010). In the same study, younger dogs had significantly higher levels of messenger RNA (mRNA) for IL8R, L-selectin, and interleukin-1β-converting enzyme. While this indicates a generally depressed innate immune response in older dogs, the ability to fight infections was not assessed in the study.
Dietary modifications also can influence the immune system in elderly humans (Lesourd, 1997; Lesourd and Mazari, 1999). However, several ingredients have not had the desired beneficial immune effects although their effects in animal studies have been generally beneficial. In humans, dietary inclusions of certain ingredients including β-carotene (Santos et al., 1997) or black-currant seed oil rich in both gamma-linolenic (18:3n-6) and alpha-linolenic (18:3n-3) acids (Wu et al., 1999) have not been reported to have significant effects on the immune system in the elderly. In contrast, vitamin E (800 mg dl-α-tocopheryl acetate) appeared to enhance cell-mediated immunity in healthy elders (Meydani et al., 1990). Immunosenescence in canines can be modulated by caloric restriction (Greeley et al., 2006), and there is an important role for nutrition in regulating immune function (Sheffy and Williams, 1981). Hall et al. (2011) reported that a diet fortified with vitamins C and E and added fish oil reduced the pro-inflammatory markers, including nuclear factor kappa B (NFκB), toll-like receptors 2 and 4 (TLR-2 and TLR-4), cyclooxygenase-2, and myeloperoxidase (MPO) in the neutrophils isolated from adult and aged dogs. Consumption of high concentrations of α-tocopheryl acetate in elderly healthy dogs resulted in higher percentages of CD8+cells when compared to those consuming low concentrations (Hall et al., 2003). These studies indicate an important role of nutrition in regulating the immune function in aging.
Renal Function
A reduction in age-associated kidney function in humans has been well established (see Abdel-Rahman and Okusa, 2014, for review). In addition to a reduction in renal mass during aging (Mulder and Hillen, 2001), morphological changes in the glomeruli are associated with kidney dysfunction, including decreases in glomerular filtration rate (GFR) and renal blood flow (Anderson and Brenner, 1986; Weinstein and Anderson, 2010). Similarly, a reduction in renal function with age has also been reported in cats (Hall et al., 2014) and dogs (Hall et al., 2015). In canines, chronic renal failure (CRF) is the most common form of renal disease; while CRF may occur at all ages, its incidence increases with age (Rubin, 1997). Inflammation is associated with a decline in renal function. Costa et al. (2013) reported impaired renal function in older rats (18 months old) and significantly higher urea and creatinine as well as interferon gamma (INFγ) when compared to young rats (two months). Increased pro-inflammatory cytokines and chemokines (CCLs), including CCL3, CCL4, CCL5, CD80, TNF-α, and IL-12b are upregulated in aging rat kidney when compared to young (Xi et al., 2014). Other pro-inflammatory mediators in kidney dysfunction in aging include NFκB (Moreno et al., 2011). In canines, there was an increased expression of the cytokines IL-1α, IL-1β, transforming growth factor beta (TGF-β), and the enzyme 5-lipoxygenase (5-LO) in the venous whole blood of dogs with renal disease (Nentwig et al., 2016). There was a significant relation between serum CRP concentrations and kidney function, which indicates an important role for CRP in the pathogenesis of naturally occurring canine renal disease (Raila et al., 2011). Median CRP concentration of miniature schnauzer dogs was slightly higher than that of other breeds of dogs (Wong et al., 2011), indicating that breed specificity may also be an important factor to consider when assessing inflammation in dogs. In cats, chronic kidney disease (CKD) is the most common metabolic disease of aged domesticated cats (>12 years of age; Brown et al. (2016). Inflammation contributes to the progression of renal fibrosis in CKD in cats; when urine cytokine levels in both healthy and CKD cats were compared, significantly higher levels of IL-8 and transforming growth factor-β1 (TGF-β1) concomitant with lower vascular endothelial growth factor (VEGF) levels were reported (Habenicht et al., 2013).
A common aging effect shared by cats, dogs, and humans is declining renal function (see Panickar and Jewell, 2015 for review; Hall et al.; Hall et al., 2016a; Hall et al., 2016b). There are multiple observational studies in humans that show diets rich in fruits and vegetables reduce the risk of chronic kidney disease (Jain and Reilly, 2014). There is also a reduction in renal function when foods high in pro-inflammatory ingredients are compared to foods with high anti-inflammatory dietary ingredients; the pro-inflammatory regimen was also associated with increased systemic inflammation (Xu et al., 2015). Therefore, it is reasonable to conclude that inflammation is one pathway through which kidney function is influenced and foods that reduce inflammation may be beneficial in reducing age-associated declines in kidney function.
Nutritional intervention to reduce kidney dysfunction is important in maintaining kidney health. In evaluating the effect of anti-inflammatory ingredients and botanicals on early intervention to change age-associated declines in renal function, a food rich in antioxidants (alpha-lipoic acid, vitamins E and C), fish oil, and botanicals (fruits and vegetables) were fed to dogs for a period of six months. Dogs that did not receive the renal protective food (RPF) but were maintained on their owner’s choice food had numerically increased blood urea nitrogen (BUN) and symmetric dimethyl arginine (SDMA), while those that received the RPF decreased BUN and SDMA. The control dogs did not change blood creatinine concentration, whereas the RPF fed dogs declined in circulating creatinine concentration. These improvements in markers of renal function in geriatric dogs show the benefit of anti-inflammatory ingredients and botanicals (Hall et al., 2016b). A major role of the kidneys is to also maintain phosphorus homeostasis in the body. In general, in dogs with chronic renal failure the combination of low protein and low phosphorus in the diet appears to be beneficial. In addition, several ingredients have been demonstrated to attenuate stress associated with kidney function in aging as well as in nonaging-related kidney dysfunction. Table 21.1 provides a list of anti-inflammatory botanical extracts that may prove beneficial as nephroprotective agents when included in the diet. In cats, similar to what was observed in dogs, an RPF enriched with antioxidants (vitamins E and C), fish oil, L-carnitine, and botanicals (vegetables) had an improvement in renal function when compared to cats that did not receive nutritional intervention with the RPF. In this study, the control cats had no change in BUN and increased SDMA concentration while urine specific gravity decreased. In contrast, cats fed the RPF saw reduced concentrations of BUN, creatinine, and SDMA. Like the dog, these improvements in markers of renal function in the geriatric cats show the benefits of anti-inflammatory ingredients and botanicals (Hall et al., 2016a). Anorexia or hyporexia is a common problem in cats with CKD and may lead to cats being fed suboptimal diets for their disease (Markovich et al., 2015), indicating an importance of nutrition in cats with kidney disease.
Table 21.1
Anti-inflammatory and Immune-Modulating Effects of Some Herbal and Plant Extracts in Both In Vivo and In Vitro Models of Inflammatory Conditions
| Bioactive Component or Extract | Class of Compound | Efficacy in Health Conditions | Potential Anti-Inflammatory Mechanism | References |
| β-caryophyllene (clove oil, rosemary, hops, basil) | Sesquiterpene | Neuroprotective, nephroprotective, immune modulation | Inhibits pathways due to activation of toll-like receptor complex CD14/TLR4/MD2; reduces pro-inflammatory cytokines, including IL-1β, TNF-α, IL-6; exhibits synergy with μ-opioid receptor–dependent pathways; activates peroxisome proliferator-activated receptor gamma (PPARγ) pathway | Bento et al., 2011; Horváth et al., 2012; Guo et al., 2014; Sharma et al., 2016 |
| Eugenol (clove) | Phenylpropene | Inflammation, nephroprotective | Inhibits IL-1β, IL-6, and matrix metalloproteinase 9 (MMP-9); inhibits mouse paw edema induced by carrageenan; anti-inflammatory effect on gentamycin-induced nephrotoxicity in rat kidney | Valacchi et al., 2009; Said 2011; Taher et al., 2015 |
| Zingerone, shogaols, and gingerols (ginger extract) | Guaiacol (zingerone) | Aging, nephroprotective | Zingerone partially prevented age-related decline in PPAR expression and suppressed pro-inflammatory NFκB activity in rats; gingerol induces nephroprotective effect on gentamycin-mediated nephropathy and reduces IL-2, TNF-α, and IFN-γ. | Chung et al., 2009; Kim et al., 2010; Rodrigues et al., 2014 |
| Proanthocyanidins (grape seed extract, cinnamon extract) | Polyphenol | Cognition, nephroprotective | Grape seed extract ameliorates renal injury in type 2 diabetic rats; proanthocyanidins from persimmon peel reduced inflammation in streptozotocin-induced diabetic rats; proanthocyanidin-rich fraction obtained from the bark of Croton celtidifolius Baill has anti-inflammatory as well as beneficial effects on cognitive functions in rats | Lee et al., 2007; Moreira et al., 2010; Panickar, 2014; Bao et al., 2015 |
| Suppressed NFkB and activator protein 1 pathways; prostaglandin E2 (PGE2), COX-2; reduction in cMyc, H-ras, and p53-related genes; inhibited signal transducer and activator of transcription 4 (STAT-4), mitogen-activated protein kinases (MAPKs); reduces TNF-α, IL-2, IL-4, IFN-γ, and chemokines (CXCL8, CCL2, CCL3, CCL4) | ||||
| Ellagitannin, gallocatechin, delphinidin (pomegranate extract) | Polyphenol | Nephroprotective, cognition | Urolithins, gut microbiota–derived metabolites of ellagitannins, inhibit LPS-induced inflammation in RAW 264.7 murine macrophages; punicalagin, a bioactive ellagitannin, inhibits LPS-induced inflammation in a macrophage cell line; consumption of pomegranate juice decreased inflammation and strengthened innate immunity in hemodialysis patients; improves cognitive performance in diabetic rats as well as in a mouse model of Alzheimer’s disease. | Cambay et al. 2011; Shema-Didi et al., 2012; Xu et al., 2014; Piwowarski et al., 2015; Subash et al., 2015 |
| Piperine (black pepper extract) | Alkaloid | Aging, nephroprotective | Inhibits STAT-1; reduces NFκB activation, suppresses Akt, ERKs; reduces IL-2, IL-4, IL-5, iNOS, COX-2, INF-γ | Kim and Lee, 2009; Bae et al., 2012 |
| Reduces eosinophil infiltration; decreases MMP-3, MMP-13 responses | ||||
| Rutin (buckwheat and citrus fruit rinds) | Polyphenol | Cognition | Improves cognition and reduces neuroinflammation in a mouse model of Alzheimer’s disease; Improves long-term and short-term episodic memory deficits in Wistar rats | Kwon et al., 2005; Koda et al., 2009; Javed et al., 2012; Ramalingayya et al., 2016 |
| Reduces tissue levels of IFN-γ, IL-1β, IL-4, IL-5, IL-10, IL-17; IL-31, and IL-32; reduces NFκB activation, COX-2, iNOS, TNF-α; inhibits MMP-3 | ||||
| Carnosol (rosemary extract, sage extract) | Polyphenol | Cognition | Rosmarinus officinalis leaf extract improves memory deficits in a rats; rosemary extract and carnosol both inhibited COX1 and COX2 activity in mice. | Ozarowski et al., 2013; Emami et al., 2013 |
| Oleocanthal, oleuropein, (olive oil) | Polyphenol, phenylethanoid (oleocanthal) | Nephroprotective, cognition | Dietary olive oil reduces inflammation and renal injury in mice; anti-inflammatory effects of oleuropein aglycone in neurodegeneration and peripheral inflammatory disorders | Casamenti et al., 2015; Aparicio-Soto et al., 2016 |
| Rhubarb extract,rhubarb (Rheum rhabarbarum) | Anthraquinones | Nephroprotective | Nephroprotective and anti-fibrotic activities in patients with chronic kidney disease; anthraquinones from rhubarb reduce HgCl2-induced acute renal failure in rats. | Zhang et al., 2015; Gao et al., 2016 |
| Withania somnifera | Tropine and cuscohygrine (alkaloid also found in cocoa), withaferin A (steroidal lactone) | Nephroprotective, cognition | Oral administration of W. somnifera significantly protected against bromobenzene-induced nephrotoxicity and renal dysfunction in rats. W. somnifera extract improved cognitive and psychomotor performance in healthy male subjects. | Vedi et al., 2014; Pingali et al., 2014 |
| Magnolol (Magnolia officinalis) | Lignan | Mobility | Anti-arthritic effects of magnolol reported in a Mycobacterium butyricum-induced arthritis model in rats | Wang et al., 2012 |
| Fenugreek extract,fenugreek (Trigonella foenum-graecum L) | Alkaloids, flavonoids | Nephroprotective | Fenugreek oil reduced renal toxicity in alloxan-induced diabetic rats. | Hamden et al., 2010; Yadav & Baquer, 2014 |
| Black cumin (Nigella sativa) | Thymoquinone, tannins | Nephroprotective, cognition | N. sativa administration antagonized paracetamol-induced kidney pathological damage, improved cognition in healthy adolescent males as well as in elderly human subjects. | Bin Sayeed et al., 2013, 2014; Canayakin et al., 2016 |
| Mangosteen (Garcinia mangostana) | Xanthones (gartanin and α-mangostin) | Cognition, nephroprotective, mobility | Mangosteen attenuated the deficit in spatial memory retrieval in a mouse model of Alzheimer’s disease. α-mangosteen had a renoprotective effect in a cisplatin-induced model of nephrotoxicity in rats. Isogarcinol, isolated from Garcinia mangostana, reduced inflammation in collagen-induced arthritis in animals. | Pérez-Rojas et al., 2009; Huang et al., 2014; Fu et al., 2014 |
| Silibinin (milk thistle: Silybum marianum) | Polyphenol | Mobility, cognition, nephroprotective | Silymarin, primarily made of three isomers—silybin, silydianin, and silychristin—exhibited significant anti-inflammatory and antiarthritic activities in the papaya latex–induced model of inflammation in rats; silymarin significantly reversed high-fat diet-induced cognitive deficits in mice; silymarin significant attenuated the nephrotoxic effects in a rat model of kidney stress. | Gupta et al., 2000; Neha et al., 2014; Alcaraz-Contreras et al., 2016 |
| Scutellaria baicalensis and Oroxylum indicum (Indian trumpetflower) | Baicalein (flavonoid) | Nephroprotective, cognitive, mobility | Baicalein attenuated kidney injury induced by myocardial ischemia and reperfusion in rats; baicalein inhibited inflammatory process through inactivation of NF-κB and MAPK signal pathways to exert antifibrotic actions in obstructive kidney disease in mice; baicalein prevented spatial learning and memory retention deficits following whole brain irradiation in mice; baicalein attenuated expression of MMPs and ameliorated cartilage damage in rabbit model of osteoarthritis. | Oh et al., 2013; Chen et al., 2015; Wang et al., 2015; Lai et al., 2016 |
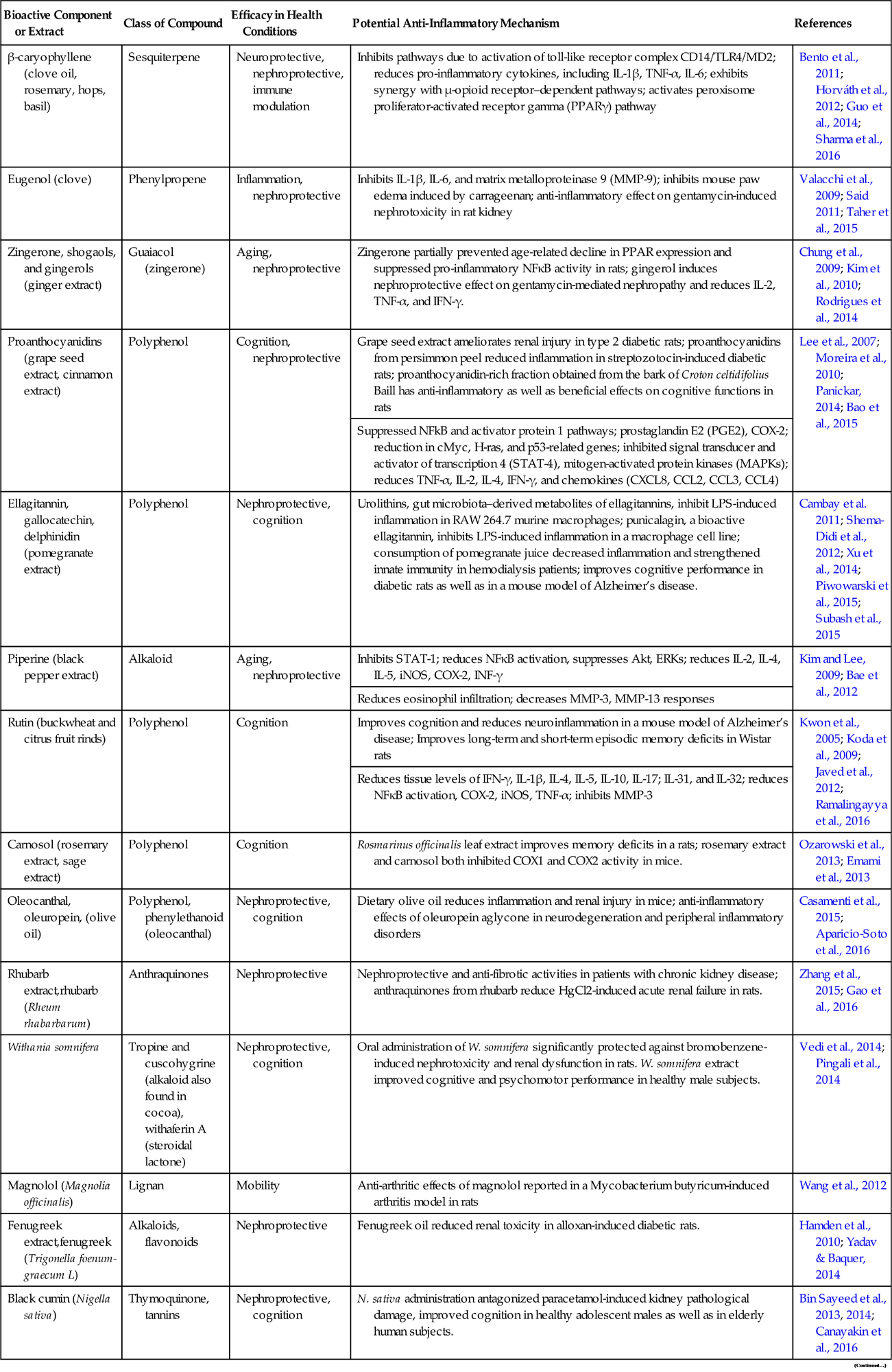
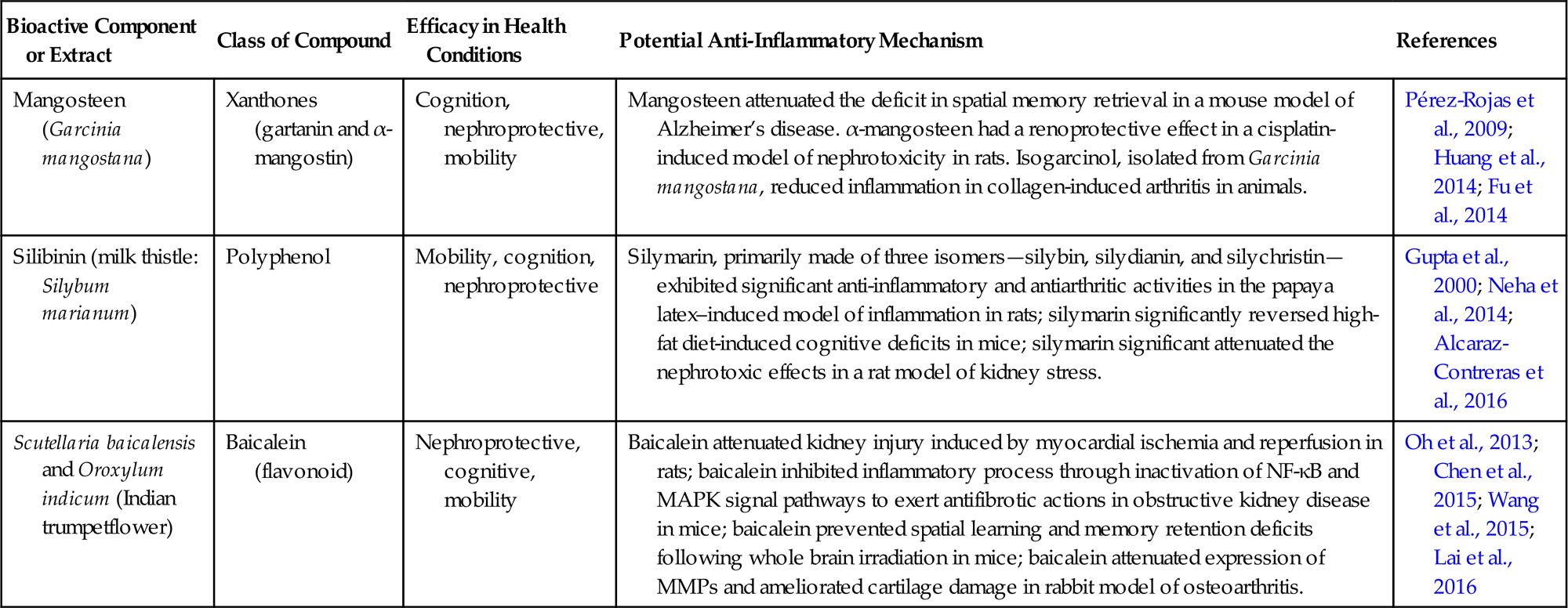
Source: Table modified from Panickar, K.S., 2014. Anti-inflammatory properties of botanical extracts contribute to their protective effects in brain edema in cerebral ischemia. In: Watson, R., Preedy, V. (Eds), Bioactive Nutriceuticals and Dietary Supplements in Neurological and Brain Disease: Prevention and Therapy, Academic Press, New York, pp. 3−15.
The nephroprotective effects of bioactive components from dietary ingredients, including botanical extracts, in rodent studies have been reported. In rats made obese with a high-fat diet, lycopene, a major component of tomato, reduced TNF-α levels in the kidney when supplemented in the high-fat diet (Pierine et al., 2014). Anti-inflammatory effects of lycopene have been reported in the kidney of obese rats (Pierine et al., 2014), in diabetic nephropathy in mice (Guo et al., 2015), and in contrast-induced nephropathy in rats (Buyuklu et al., 2015). Inclusion of polyphenols, which generally have anti-inflammatory effects, has also been shown to have a nephroprotective effect. In a mouse model of diabetes, the low-molecular-weight polyphenol oligonol, derived from lychee fruit, reduced diabetes-induced increase in advanced glycation end product (AGE) formation and apoptosis in the kidneys (Park et al., 2014). A polyphenol-rich extract from amla (Emblica officinalis Gaertn) reduced the expression of renal nuclear factor kappa B (NFκB), inhibitory κB in cytoplasm, inducible nitric oxide synthase (iNOS), and cyclooxygenase 2 (COX-2) protein levels that were elevated in aged rats (Yokozawa et al., 2007).
In summary, it is apparent that a diet that includes anti-inflammatory and specific botanically active ingredients can reduce or reverse the decline in kidney function associated with aging. This effectiveness is likely due in part to the known beneficial effect of the anti-inflammatory ingredients but may also be the result of a specific action of individual botanical compounds on the kidney.
Cognitive Function
Cognitive function declines with aging, but it is not uniform; some people experience very little cognitive decline whereas others suffer from mild to moderate to severe decline in some components of cognitive function. The causes or mechanisms of this decline are not clear, but there is evidence to indicate a role for inflammation in reducing cognitive ability. In aged rats that received a single injection of lipopolysaccharide (LPS), there was an impaired reversal of learning and attentional shifts but not effect on discrimination learning (Culley et al., 2014). Further, in this study, although an increase in monocyte chemoattractant protein 1 (MCP-1) was found elevated 2 h after LPS administration, the levels returned to normal at the time of testing. Whether the increase and subsequent decrease in TNF and CCL2 following LPS injection in the rats had initiated a cascade of events that affected subsequent cognitive function is not clear but is a possibility. Increased levels of TNFα and IL1β were reported in the 24-month group (older group) when compared to the young group of rats (Gocmez et al., 2016). In another study in aged rats, following laparotomy (abdominal cavity surgery) memory and learning functions were impaired and there was a significant upregulation of TNF-α, interleukin (IL)-1β, IL-4, and IL-6 in the hippocampal tissues. However, intracisternal administration of the TNF-α receptor antagonist R-7050 during surgery attenuated these defects in cognitive function and inhibited the production of the pro-inflammatory cytokines (Ma et al., 2015).
In human subjects, a study reported that a pro-inflammatory diet at midlife might be associated with subsequent lower cognitive functioning (Kesse-Guyot et al., 2016). In the Singapore Longitudinal Aging Study, significant associations of soluble IL-2 receptor alpha chain, soluble tumor necrosis factor receptor 2, and soluble glycoprotein 130 were found along with cognitive impairment in community-dwelling older persons (Gao et al., 2016). In the Berlin Aging Study II, levels of IL-6, IL-10, and CRP were inversely associated with executive function and processing speed, but IL-6 to IL-10 ratio was not predictive for executive function and processing speed (Tegeler et al., 2016). Further, in the same study, no associations were found between inflammatory markers and verbal episodic memory. The underlying mechanisms or the neuroanatomical correlates on why certain inflammatory markers are associated with certain components of memory are not clear. In nondemented subjects aged 70–90 years, higher levels of serum macrophage inhibitory cytokine-1 (MIC-1), also called growth differentiation factor 15 (GDF15), were associated with lower global cognition (Fuchs et al., 2013). A higher intracellular cytokine production of IL-1β and IL-6 by activated monocytes were predictive of lower cognitive performance in working memory in healthy older individuals aged 55–70 years (Simpson et al., 2013). In a prospective population-based cohort study with data collected over 20 years, it was reported that higher serum CRP and IL-6 were associated with a likelihood of cognitive impairment (Wichmann et al., 2014). Nascimento et al. (2014) reported an association of pro-inflammatory cytokines, including TNF-α and IL6, with mild cognitive impairment in elderly humans. Such elevated levels of the pro-inflammatory cytokines, however, could be reduced with physical exercise, which also correlated with subsequent positive effects on cognition. Taken together, these studies indicate that an increased risk of cognitive decline is associated with age-related inflammatory conditions.
In canines, an age-dependent decline is seen in learning and memory, and neuropathological changes are also observed in canines that are generally similar to those seen in normal human aging or in early Alzheimer’s disease (Head, 2011; Vite and Head, 2014; Schütt et al., 2016). Cognitive dysfunction syndrome has also been described in canines and felines (Landsberg et al., 2012; Bosch et al., 2013). Interferon gamma (IFN-γ) expression was increased in the homogenates of dentate gyrus, a region of the limbic system involved in cognition, in aged dogs when compared to adult dogs (Hwang et al., 2008). A battery of cognitive tests were then administered to dogs that were divided into young (1–4 years), middle-aged (5–8 years), cognitively unimpaired aged (≥9 years), and cognitively impaired aged (≥9 years). The younger dogs (<9 years) successfully located the food more quickly in a food-seeking task and with more success than the aged groups (≥9 years) (González-Martínez et al., 2013). In another study with pet dogs. there were no significant effects of age on learning or retention tasks, but older dogs (≥8 years) were significantly impaired on the reversal learning task when compared with younger ones (<8 years). In addition, the trial response latency was significantly increased in aged dogs in both the initial and reversal learning tasks but not on the retention task (Mongillo et al., 2013). These studies indicate that pro-inflammatory markers are associated with a decline in cognitive function, but the facets of cognitive components affected by various inflammatory markers are yet unclear.
Diet-induced improvement in cognitive function has been demonstrated and it appears that more research is warranted. In the Whitehall II prospective cohort study, a dietary pattern with a higher intake of red and processed meat, peas, legumes and fried food, and lower intake of whole grains was associated with higher inflammatory markers and accelerated cognitive decline at older ages (Ozawa et al., 2016). Intervention studies using eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), fatty acids which have anti-inflammatory effects, to adults with mild cognitive impairment or age-related cognitive impairment have shown beneficial effects although no differences in cognitive decline between treated and nontreated participants were observed in healthy older adults (Cederholm et al., 2013). In a population-based cohort of nondemented Asian subjects aged 60–93 years, when the authors compared the Mini-Mental State Examination (MMSE) scores for three categories of regular curry (curcumin) consumption they found that those who consumed curry “occasionally” and “often or very often” had significantly better MMSE scores than did subjects who “never or rarely” consumed curry (Ng et al., 2006). The anti-inflammatory effects of curcumin have been well established in several studies (for reviews, see Boyanapalli and Tony Kong, 2015; Ghosh et al., 2015; Sahebkar et al., 2016).
In canines, higher serum vitamin E concentrations from vitamin E supplemented food correlated with improved performance on landmark-discrimination tasks in aged dogs (Ikeda-Douglas et al., 2004). Although vitamin E (generally tocopherols) has been reported to have antioxidant effects, their anti-inflammatory effects have also been reported (Betti et al., 2011; England et al., 2012). Whether the improvement in cognitive tasks was a consequence of their anti-inflammatory actions or a combination of antioxidant and anti-inflammatory actions is not clear but is a possibility. A combination of antioxidant and mitochondrial cofactor improved the performance of aged dogs in a three-choice size-discrimination task (Siwak et al., 2005). Canine puppies fed a diet rich in DHA (and with higher concentrations of vitamin E, taurine, choline, and L-carnitine) performed significantly better on cognitive tasks when compared to moderate and low DHA groups (Zicker et al., 2012). In addition to the anti-inflammatory actions of vitamin E, DHA also has been reported to have anti-inflammatory actions in beagles (Hall et al., 2011). Canines supplemented with medium-chain triglycerides (MCT) showed improved memory in some but not all of the cognitive tasks administered in aged dogs (Pan et al., 2010). The mechanisms underlying the improvement in cognitive functions in the previously mentioned studies is not clear, but certainly ingredients with anti-inflammatory potential may be important in attenuating cognitive decline.
In adult rats, anthocyanins reversed the D-galactose–induced neuroinflammation-mediated cognitive impairment (Rehman et al., 2016). In aged rats, dietary supplementation with the polyphenol-rich açaí pulps (Euterpe oleracea Mart. and Euterpe precatoria Mart.) improved cognition in aged rats and concomitantly reduced levels of TNF-α were also reported in the serum (Carey et al., 2015). In mice, supplementing a diet with resveratrol reduced the LPS-related neuroinflammation and deficits in working memory in aged mice (Abraham and Johnson, 2009). Similarly, resveratrol was effective in preventing cognitive deficit in aged rats by inhibiting the production of inflammatory cytokines (Gocmez et al., 2016). The inclusion of mushroom (Agaricus bisporus), which has anti-inflammatory effects, in the daily diet of aged rats had beneficial effects on age-related deficits in cognitive and motor function (Thangthaeng et al., 2015). Supplementation with blueberry polyphenols attenuated the kainic acid–induced increase in the expression of IL-1β, TNF-α, and NFκB, and expression of insulin-like growth factor 1 (IGF-1) and a concomitant reduction in learning impairments following the neurotoxic insult (Shukitt-Hale et al., 2008). In addition, the effects of polyphenol-rich extracts from grape, strawberry, blackberry, and plum reportedly have beneficial effects in cognition in rodent models (Shukitt-Hale et al., 2006; Shukitt-Hale et al., 2009; Cherniack, 2012). Table 21.1 lists some natural compounds that may provide beneficial health effects by reducing inflammation and may also benefit cognition. In short, the studies mentioned indicate that dietary ingredients, in particular botanical extracts with anti-inflammatory properties, may be useful in improving several components of cognitive function.
Conclusion
Aging-associated increases in low-grade systemic inflammation could contribute to the pathogenesis of various chronic conditions in aging, including kidney dysfunction, diminished cognition, and a reduction in an effective immune function. The causal factors for the increased systemic low-grade inflammation are not clear but likely multifactorial. Strategies to effectively combat aging-associated inflammation have included nutrition, drugs, lifestyle changes, and recently stem cell therapies to modulate the immune system. Nutritional intervention is a particularly important strategy to lower systemic inflammation in aging, and evidence for this comes from laboratory animal studies, clinical studies, and epidemiological studies. Dietary factors, including a polyphenol-rich diet from fruits, vegetables, and herbs, as well as a diet rich in ω-3 fatty acids and some vitamins appear to have beneficial effects or the potential to reduce systemic inflammation in aging. In general, there appears to be a health benefit in reducing systemic inflammation in aging. Given the complexity and breadth of the inflammatory profile, in particular in aging, it is imperative that the anti-inflammatory and immune-modulating properties of herbal or plant extracts be investigated further as part of the dietary regimen.
Conflict of Interest
The authors are employees at the Science and Technology Center of the Hillspet Nutrition Center, a Colgate-Palmolive company.
