Lecoq explored Samarsky’s ore, Sm’s fine lines to see,
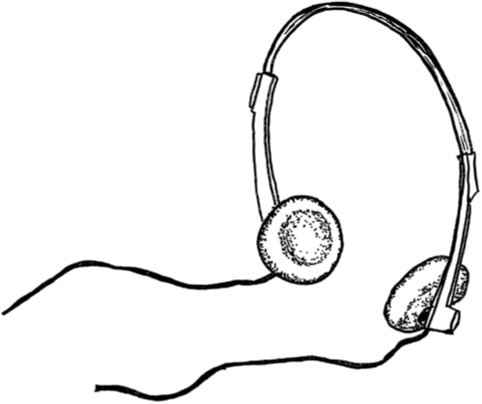
Pre-NIB its Co5 held sway, thus headphones came to be.
Keeps best when stored ’neath argon’s layer,

Lecoq explored Samarsky’s ore, Sm’s fine lines to see,
Pre-NIB its Co5 held sway, thus headphones came to be.
Keeps best when stored ’neath argon’s layer,
ess-emms
SIXTH IN THE LANTHANOID SERIES
Samarium was discovered in 1879 when French chemist Paul-Émile Lecoq de Boisbaudran (1838–1912) observed some previously unassigned spectral lines in the atomic spectrum produced by a sample he had skillfully extracted from the mineral samarskite. Lecoq de Boisbaudran named the new element after samarskite. Samarskite had been named after Colonel Vasili Samarsky-Bykhovets (1803–1870), the chief of staff of the Russian Corps of Mining Engineers between 1845 and 1861. Colonel Samarsky achieved the rare and highly desirable distinction of having an element named in his honor, albeit indirectly.
pree-nib see-oh-five
Samarium’s alloys with cobalt (for example, SmCo5) produce magnets that are 10,000 times stronger than magnets made from iron. Samarium-cobalt magnets were instrumental in the miniaturization of headphones (before the later development of the even stronger NIB10 magnets). This innovation ushered in the age of the personal stereo in the early 1980s.
[’neath: preposition — underneath]
Samarium oxidizes in moist air and even when stored under mineral oil. It also ignites spontaneously when heated above 150°C. Storing samarium in a sealed container under an inert gas, such as argon, is the best way to preserve a pure sample of the metal.
eye-arrs
Glass containing samarium oxide (Sm2O3) absorbs IR rays.
one-four-nine
The isotope samarium-149 is an effective neutron absorber. It’s added to control rods that regulate the fission reactions within the cores of nuclear reactors. (See “Cadmium, Cd” on page 141 for more on how control rods work.)