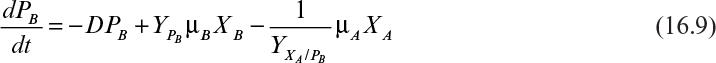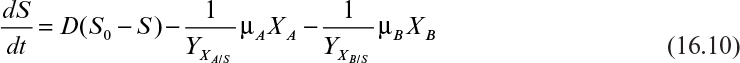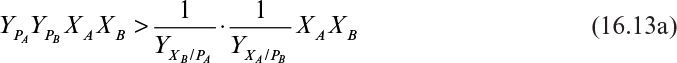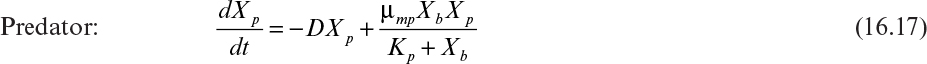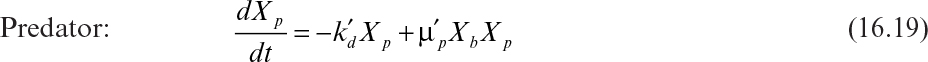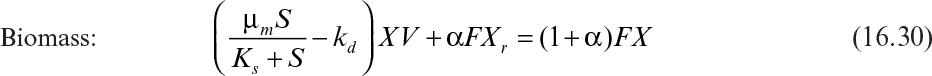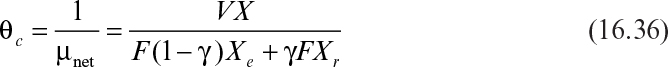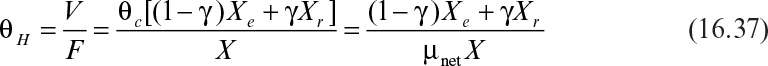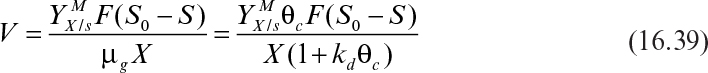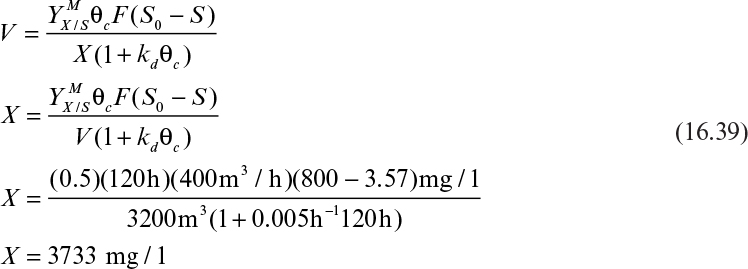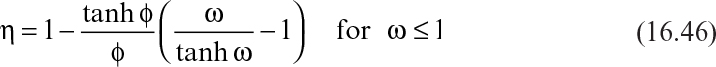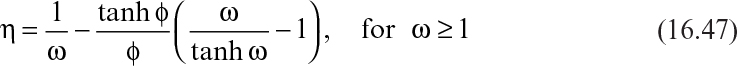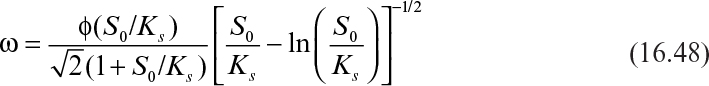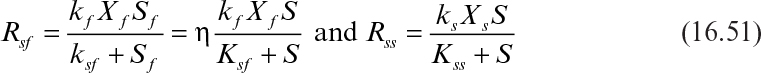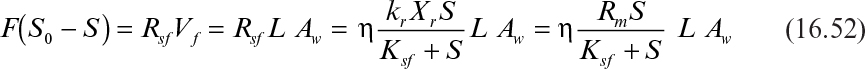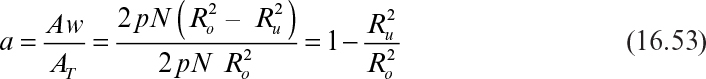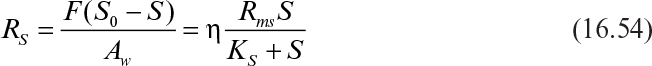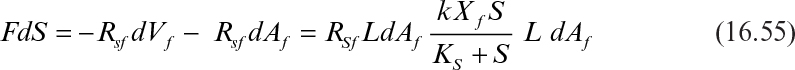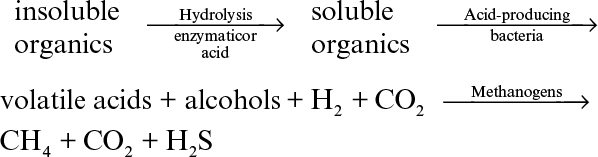16. Bioprocesses Utilizing Mixed Cultures
The dynamics of mixed cultures (or multiple species in culture) are important considerations in some commercial fermentations. They are critical to understanding the response of many ecological systems to stress. The use of organisms with recombinant DNA has added another dimension to our consideration of how cells within a population interact with each other.
Many food fermentations, such as cheese manufacture, depend on multiple interacting species. The biological treatment of wastewaters relies on an undefined complex mixture of microorganisms. The ratio of various species in the treatment process is critical; sudden shifts in the composition of the population can lead to failure of the unit to meet its objectives.
In all natural environments, cells exist in potentially mixed populations. Understanding how these cells interact with one another is critical to understanding the natural cycles for the elements (e.g., C, N, and S), the response of ecosystems to environmental challenges (for example, acid rain), and the rate and extent of degradation of chemicals introduced into such ecosystems.
As discussed in Chapter 14, “Utilizing Genetically Engineered Organisms,” many populations with recombinant DNA behave as mixed-culture systems. Some members of the population will lose or modify the inserted gene (often carried on a plasmid). Although a single species is present, the formation of mutant or plasmidless cells leads to a distinct subpopulation. The interaction of the subpopulation with the original population follows the principles developed in this chapter for mixed populations.
16.1. Major Classes of Interactions in Mixed Cultures
The major interactions between two organisms in a mixed culture are competition, neutralism, mutualism, commensalism, amensalism, and prey–predator interactions. Table 16.1 summarizes these interactions.
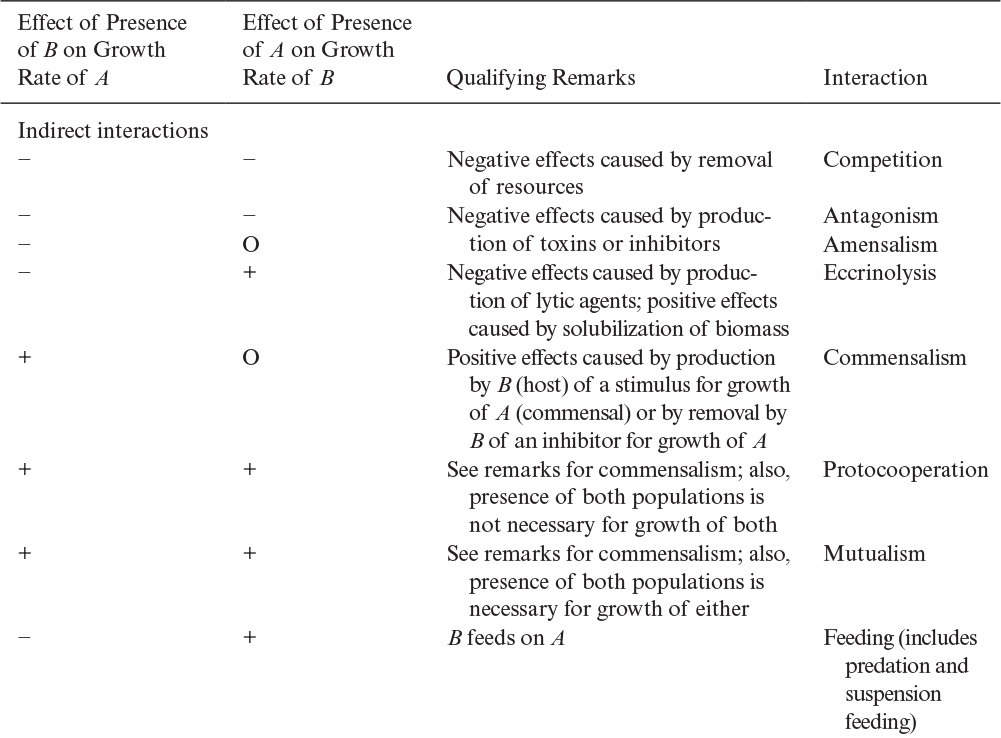

TABLE 16.1. Scheme of Classification of Binary Population Interactions (Roles of A and B May Be Reversed)*
Competition is an indirect interaction between two populations that has negative effects on both. In competition, each population competes for the same substrate. Two populations or microorganisms with similar nutrient requirements usually compete for a number of common, required nutrients when grown together.
The outcome of competition between two species for the same growth-limiting substrate in an open system (e.g., a chemostat) is determined by the specific growth-rate-limiting substrate concentration relationship. Two different cases can be distinguished in a mixed culture of two competing species (Figure 16.1):
• μa is always greater than μb. The organisms with the fastest growth rate will displace the others from the culture. This is known as the exclusion principle.
• Crossover in μ–S relationship. In this case, the faster-growing organism is determined by the dilution rate. Depending on the dilution rate, three different cases may be identified:
• At the crossover point D = μx; S = Sx, two species could be maintained in a chemostat at D = μx. However, this is an unstable operating point.
• If D > μx, then μa > μb, and B will be washed out; A will dominate.
• If D < μx, then μa > μu, and A will be washed out; B will dominate.
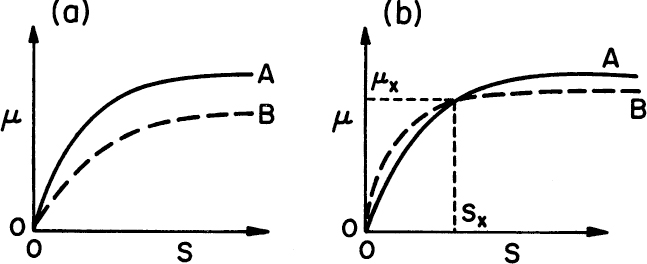
Figure 16.1. μ–S relationship for two competing species (A and B) in a mixed culture. Case (a) corresponds to μmA < μmB and KSA ≤ KSB, while case (b) corresponds to a case where μmA > μmB, but KSA > KSB. In case (b), the growth curves cross and A and B could potentially coexist at D = μx and S = Sx in a chemostat.
In a batch system, both species would exist in culture media. The ratio of number density of species at a given time will be determined by the relative magnitudes of the specific growth rates and the initial concentrations of cells.
Neutralism is an interaction where neither population is affected by the presence of the other. That is, there is no change in the growth rate of either organism due to the presence of the other. Neutralism is relatively rare. One example of neutralism is the growth of yogurt starter strains of Streptococcus and Lactobacillus in a chemostat. The total counts of these two species at a dilution rate of 0.4 h–1 are quite similar whether the populations are cultured separately or together. Neutralism may occur in special environments where each species consumes different limiting substrates and neither species is affected by the end products of the other.
Mutualism and protocooperation are more common than neutralism and may involve different mechanisms. In both cases, the presence of each population has a positive effect on the other. For mutualism, the interaction is essential to the survival of both species. In protocooperation, the interaction is nonessential. One mechanism is the mutual exchange of required substances or the removal of toxic end products by each organism. The metabolisms of partner populations must be complementary to yield a mutualistic interaction. An example is the growth of a phenylalanine-requiring strain of Lactobacillus and a folic-acid-requiring strain of Streptococcus in a mixed culture. Exchange of the growth factors phenylalanine and folic acid produced by partner organisms helps each organism to grow in a mixed culture, while separate pure cultures exhibit no growth. Another example of mutualistic interaction exists between aerobic bacteria and photosynthetic algae. Bacteria use oxygen and carbohydrate for growth and produce CO2 and H2O. The algae convert CO2 to carbohydrates and liberate oxygen in the presence of sunlight.
Note that symbiosis and mutualism are not the same. Symbiosis implies a relationship when two organisms live together. A symbiotic relationship may be mutualistic, but it may also be neutralistic, parasitic, commensalistic, and so on.
Commensalism is an interaction in which one population is positively affected by the presence of the other. However, the second population is not affected by the presence of the first population. Various mechanisms may yield a commensal interaction. Two common mechanisms are the following:
• Type 1: The second population produces a required nutrient or growth factor for the first population.
• Type 2: The second population removes a substance from the medium that is toxic to the first population.
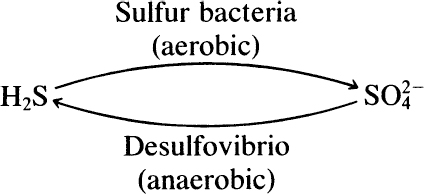
An example of the first type of commensal interaction is the production of H2S by Desulfovibrio (through the reduction of ![]() ), which is used as an energy source by sulfur bacteria (Figure 16.2).
), which is used as an energy source by sulfur bacteria (Figure 16.2).
An example of a type 2 commensal interaction is the removal of lactic acid by the fungus Geotrichum candidum, which allows the growth of Streptococcus lactis. This interaction is utilized in cheese making using S. lactis. Lactic acid produced by S. lactis inhibits the growth of the bacteria. The fungus metabolizes lactic acid and improves the growth conditions for S. lactis.
Amensalism is the opposite of commensalism. In amensalism, population A is negatively affected by the presence of the other population (B). However, population B is not affected by the presence of population A. Various amensal interaction mechanisms are possible. Two common mechanisms are the following:
• Type 1: Population B produces a toxic substance that inhibits the growth of population A.
• Type 2: Population B removes essential nutrients from the media, thus negatively affecting the growth of population A.
One example of the first type of amensal interaction is the production of antibiotics by certain molds to inhibit the growth of others. Some microbes excrete enzymes that decompose cell-wall polymers. Such organisms destroy their competitors and also utilize the nutrients released by the lysed cells. The microbial synthesis of organic acids reduces pH and inhibits the growth of other organisms.
Predation and parasitism are interactions in which one population benefits at the expense of the other. These two interactions are distinguished by the relative size of organisms and the mechanisms involved. Predation involves the ingestion of prey by the predator organism. A good example of prey–predator interaction is the ingestion of bacteria by protozoa. This interaction is common in aerobic waste-treatment reactors such as activated sludge units. In parasitism, the host, which is usually the larger organism, is damaged by the parasite. The parasite benefits from utilization of nutrients from the host. A common example of parasitism is the destruction of microorganisms by microphages. Although the physical mechanisms in predation and parasitism differ, these two phenomena have many common features in their conceptual and mathematical descriptions.
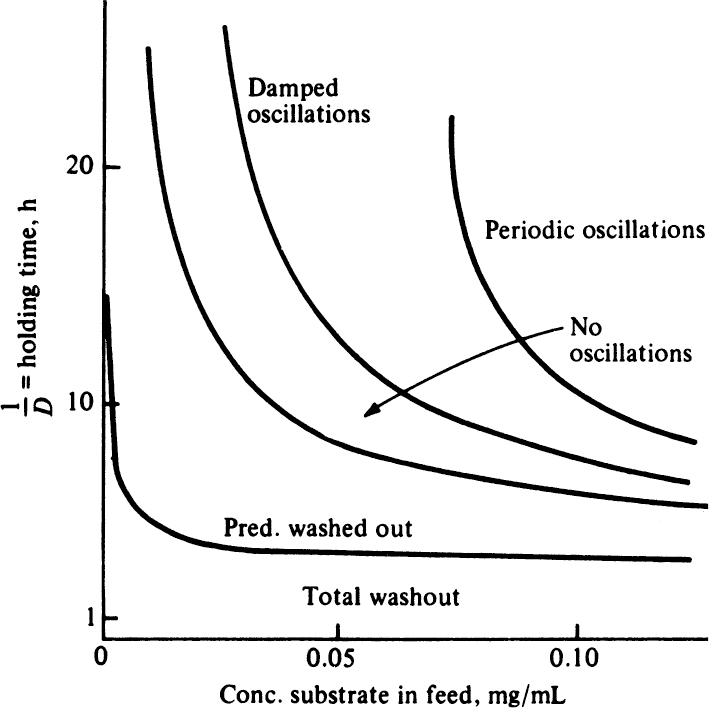
In an open system, such as a chemostat where predator–prey interactions take place, the populations of predator and prey do not necessarily reach steady state but can oscillate at certain dilution rates. At the beginning of the operation prey concentration is high, but predator concentration is low. As the predators consume prey, the number of predators increases and the prey concentration decreases. After a while, a small prey population cannot support the large predator population, and the predator population decreases while prey population increases. Depending on the dilution rate and feed substrate concentration, these oscillations may be sustained or damped or may not exist.
Finally, note that these interactions can, and often do, exist in combination. For example, A and B may compete for glucose as a nutrient, but A requires a growth factor from B to grow. In such a case, both competition and commensalism would be present.
16.2. Simple Models Describing Mixed-Culture Interactions
Multiple interacting species can give rise to very complex behavior. In some cases, coexistence of species is prohibited; in others, complex sustained oscillatory behavior may be observed. Multiple steady states are possible.
Writing the appropriate equations to describe mixed populations follows the principles we discussed in Chapter 6, “How Cells Grow.” In each case, a balance must be written for each species (organism, rate-limiting substrate, or product), and these balances will be the same as we have used previously. Although we may write chemically structured models for each organism, we confine our discussions to the use of unstructured models for each species. Even so, the population model is still structured in the sense that the whole biomass is divided into distinct subpopulations.
These equations are often applied to chemostats that mimic many ecosystems. When these equations are solved, they often yield multiple steady states. For each steady state we need to analyze its stability. An unstable steady state will never be realized in practice; using phase-plane portraits, we can explore the approach to the steady state and the determination of the presence or absence of limit cycles. It is often easier to test a steady state for its local stability (linearized stability analysis) than for its global stability.
With this background, we will consider some representative examples.
Although this coexistence is mathematically obtainable, it is not physically attainable in real systems. In real systems, the dilution rate will vary slightly with time, and, in fact, the variation will show a bias. Using an analysis more sophisticated than appropriate for this text, it can be demonstrated that one competitor will be excluded from the chemostat if the intensity of the “noise” (random fluctuations) in D and the bias of the mean of D away from Dc are not both zero. Also, it is possible for either competitor to be excluded, depending on how D varies.†
† G. N. Stephanopoulos, R. Aris, and A. G. Fredrickson, Mathematical Biosciences 45: 99, 1979.
Two competitors can coexist if we modify the conditions of the experiments. Examples of such modifications include allowing spatial heterogeneity (the system is no longer well mixed or wall growth is present) or another level of interaction is added (e.g., adding a predator or interchange of metabolites). Also, operation of the chemostat in a dynamic mode (D is a function of time) can sometimes lead to coexistence. It is also interesting to note that the use of other rate expressions (e.g., substrate inhibition) can lead to multiple crossover points and potentially multiple steady states.
This model, based on saturable substrate uptake, can be compared to the classical model developed by Lotka and Volterra almost a century ago. The Lotka–Volterro model does not consider that substrate influences the growth role of the prey and does not assume an open system, such as a chemostat. The equations describing this system follow:
The first term in equation 16.18 describes the growth of prey on substrate, and the second describes the consumption of prey by predators. The first and second terms in equation 16.19 describe the death of predator in the absence of prey and the growth of predator on prey, respectively. Yp/b is the yield of predators on prey (g/g), ![]() is the specific growth rate of prey on a soluble substrate (h–1),
is the specific growth rate of prey on a soluble substrate (h–1), ![]() is the growth rate of predator on prey (l/g-h), and
is the growth rate of predator on prey (l/g-h), and ![]() is the specific death rate of the predator (h–1)
is the specific death rate of the predator (h–1)
Equations 16.18 and 16.19 allow a steady-state solution for batch growth, where dXb/dt and dXp/dt = 0. Under these conditions, ![]() and
and ![]() .
.
By defining dimensionless variables such as
we can express equations 16.18 and 16.19 in terms of ![]() and
and ![]() :
:
Equations 16.21 and 16.22 can be solved with the initial conditions of ![]() and
and ![]() .
.
The relationship between ![]() and
and ![]() can be determined by dividing equation 16.21 by equation 16.22:
can be determined by dividing equation 16.21 by equation 16.22:
Integration of equation 16.23 yields
or
where K is an integration constant that is a function of the initial population sizes.
The phase-plane analysis of the system can be made using equation 16.24. Figure 16.4 describes the limit cycles (oscillatory trajectories) of prey–predator populations for different initial population levels.

Figure 16.4. Schematics of phase-plane portraits for prey–predator interactions. (a) Limit cycles predicted by Lotka–Volterra (soft oscillations where initial conditions determine the dynamic behavior). (b) Limit-cycle prediction using the model developed in Example 16.3 (hard oscillations where the limit cycle is independent of initial conditions). The predicted steady-state point is defined by XpF and XbF. A, B, and C represent different initial conditions.
The Lotka–Volterra model considers the exponential growth of prey species in the absence of predator and neglects the utilization of substrate by prey species according to Monod form. The Lotka–Volterra oscillations depend on initial conditions and change their amplitude and frequency in the presence of an external disturbance. These types of oscillation are called soft oscillations. The other model based on Monod rate expressions (equations 16.15, 16.16, and 16.17) explains the more stable and sustained oscillations observed in nature, which are independent of initial conditions (that is, hard oscillations).
16.3. Mixed Cultures in Nature
Mixed cultures of organisms are common in natural ecological systems. Microorganisms are involved in the natural cycles of most elements (e.g., carbon, nitrogen, oxygen, and sulfur). Simplified diagrams of the carbon and nitrogen cycles are presented in Figures 16.5 and 16.6. Organisms living in soil and aquatic environments actively participate in carbon and nitrogen cycles. For example, certain organisms fix atmospheric CO2 to form carbohydrates, while others degrade carbohydrates and release CO2 into the atmosphere. Similarly, some organisms fix atmospheric nitrogen (N2) to form ammonium and proteins, while others convert ammonium into nitrite and nitrate (nitrification), and others reduce nitrate into atmospheric nitrogen (denitrification). Sulfur-oxidizing organisms convert reduced sulfur compounds (sulfur and sulfide) into sulfate, and sulfate-reducing organisms reduce sulfate into hydrogen sulfide.
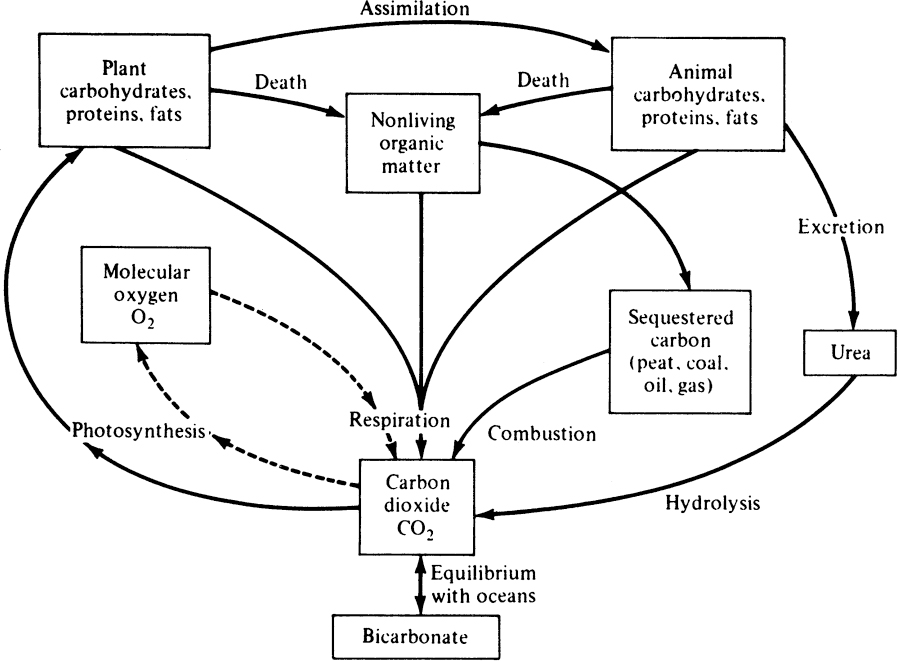
Figure 16.5. Simplified diagram of the carbon cycle. Dashed lines show the major component of the oxygen cycle, which is closely linked to the cycle of carbon. (With permission, from J. E. Bailey and D. F. Ollis, Biochemical Engineering Fundamentals, 2d ed., McGraw-Hill Book Co., New York, 1986, p. 914.)
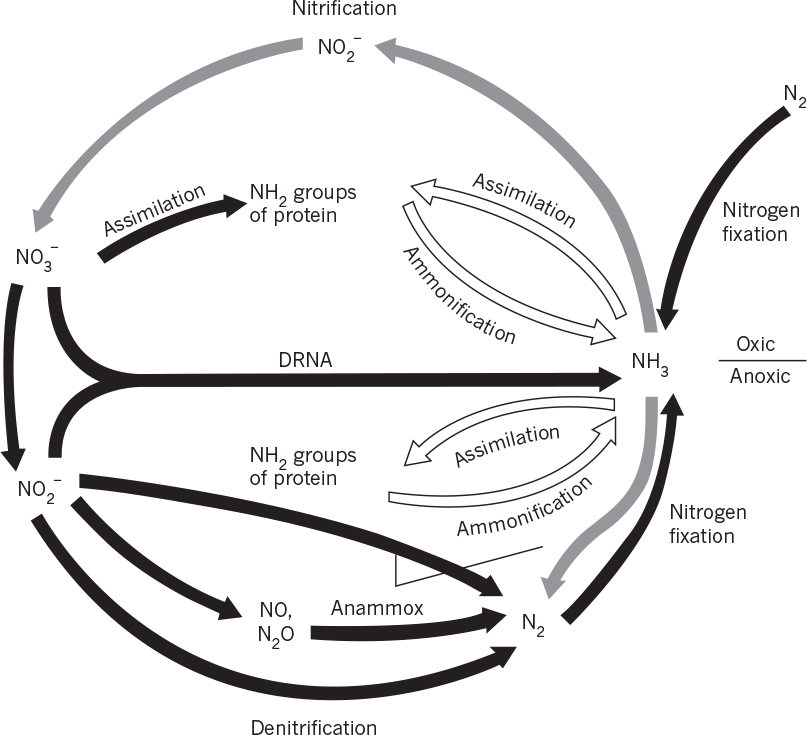
Figure 16.6. Redox cycle for nitrogen. (Brock, Thomas D.; Brock, Katherine M.; Ward, David M., Basic Microbiology with Applications, 14th ed., © 2015. Reprinted and electronically reproduced by permission of Pearson Education, Inc., New York, NY.)
The aforementioned interactions among different species take place in natural systems in a more complicated manner. The complexity of such a system is depicted in Figure 16.7 for an anaerobic environment. Understanding such complex cycles is important to understanding the capacity of natural ecosystems to degrade organic pollutants.
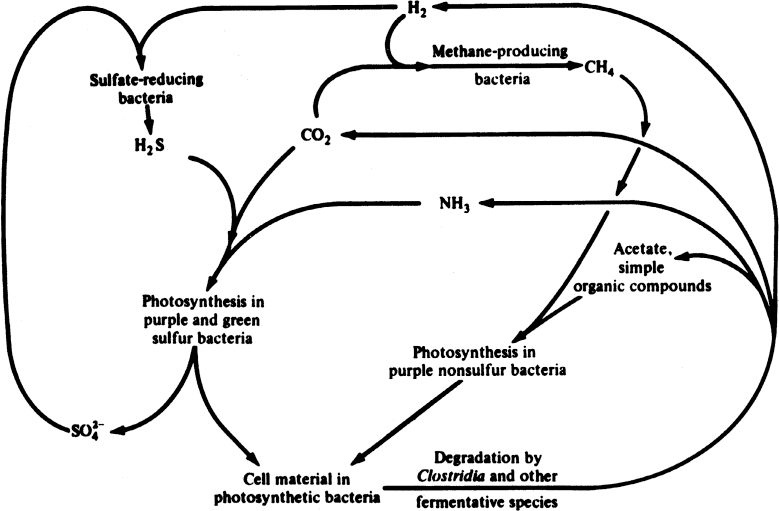
Figure 16.7. Simplified schematic of the cycles of matter in an anaerobic environment. (With permission, from J. E. Bailey and D. F. Ollis, Biochemical Engineering Fundamentals, 2d ed., McGraw-Hill Book Company, New York, 1986, p. 918.)
16.4. Industrial Utilization of Mixed Cultures
Defined mixed microbial populations are commonly used in cheese making, a good example of using mixed cultures in food production. Cheeses of various types are produced by inoculating pasteurized fresh milk with appropriate lactic acid organisms. The bacteria used for lactic acid production are various species of Streptococcus and Lactobacillus in a mixed culture. Other organisms are used to develop flavor and aroma. Among these are Brevibacterium linens, Propionibacterium shermanii, Leuconostoc sp., and Streptococcus diacetilactis. After inoculation of pasteurized milk, a protein-rich curd is precipitated by the acidity of the medium, and the liquid is drained off. The precipitated curd is allowed to age by action of bacteria or mold. Some molds used in cheese making are Penicillium camemberti and Penicillium roqueforti.
Lactic acid bacteria are also used in whiskey manufacture. Lactobacillus added to the yeast reduces pH and, therefore, the chance of contamination. Lactobacillus also contributes to the flavor and aroma of whiskey. A favorable interaction between yeast and lactic acid bacteria exists in ginger-beer fermentation.
The utilization of undefined mixed microbial cultures in waste-treatment processes is typical and unavoidable. Wastewater treatment constitutes one of the largest-scale uses of bioprocesses. Mixed cultures are also utilized in the anaerobic digestion of waste materials. Cellulase producers, acid formers, and methane producers are typical organisms involved in the anaerobic digestion of cellulosic wastes. However, attempts to encourage the growth of a particular species on waste materials have been made.
The Symba process was developed in Sweden for treating starchy wastes, particularly those from potato processing. This process utilizes Endomycopsis fibuligera for amylase production and a yeast, Candida utilis, for the utilization of sugar molecules produced from the hydrolysis of starch. Single-cell protein (SCP) is produced simultaneously with potato waste treatment.
Corn and pea wastes are also treated by a mixed culture of Trichoderma viride and Geotrichum sp. T. viride produces cellulase to break down cellulose into reduced sugar molecules, and Geotrichum produces amylases to break down starch into reduced sugar molecules. Both organisms utilize reduced sugar molecules for growth.
A mixed culture of Candida lipolytica and Candida tropicalis has been grown on hydrocarbons, n-paraffins, or gas oil for SCP production purposes in both laboratory- and pilot-scale operations. The utilization of a mixed culture of yeasts was proved to yield better product quality as compared to pure yeast strains.
Gaseous hydrocarbon substrates such as methane can be utilized by certain bacteria to produce SCP. Several experimental studies have shown that mixed cultures of methane-utilizing organisms grow faster than pure cultures.
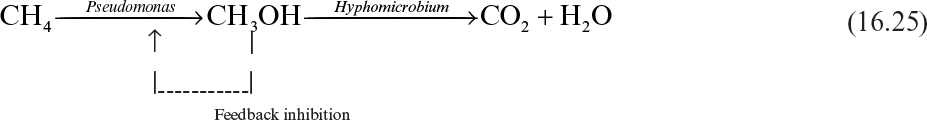
Certain methane-utilizing species of Pseudomonas oxidize methane to methanol. However, Pseudomonas is inhibited by the end product, methanol. Inclusion of a methanol-utilizing bacteria such as Hyphomicrobium into the growth medium eliminates the problem of methanol inhibition. This relationship is mutualistic in the sense that Pseudomonas supplies carbon source (CH3OH) for Hyphomicrobium, and Hyphomicrobium removes the growth inhibitor (methanol) of Pseudomonas:
16.5. Biological Waste Treatment
Biological waste treatment is an example of the industrial utilization of mixed cultures. Waste materials generated in a society can be classified in three major categories:
• Industrial wastes are produced by various industries, and waste characteristics vary greatly from one industry to another. Industrial wastes usually contain hydrocarbons, carbohydrates, alcohols, lipids, and aromatic organics. Industrial wastes are rich in carbon compounds and usually deficient in nitrogen (high C/N ratio); therefore, the biological treatment of industrial wastes usually requires supplemental addition of nitrogen compounds and other nutrients. The presence of potentially toxic compounds must be carefully considered in devising a treatment strategy.
• Domestic wastes are treated by municipalities and derive from humans and their daily activities. They include ground garbage, laundry water, excrement, and often some industrial wastes that have been sewered into the municipal system. Domestic waste varies significantly with time in terms of flow and composition due to the periodic nature of human activity (e.g., flow decreases at night when most people sleep).
• Agricultural wastes are produced by farm animals (e.g., manure) and include waste plants, such as straws. Agricultural wastes are usually carbon rich because of high cellulosic material content, although some wastes, such as poultry manure, are high in nitrogen.
Each of these waste materials has its own characteristics, and treatment methods vary depending on these characteristics.
Three major waste treatment methods are the following:
• Physical treatment includes screening, flocculation, sedimentation, filtration, and flotation, which are usually used for the removal of insoluble materials.
• Chemical treatment includes chemical oxidations (chlorination, ozonation) and chemical precipitation using CaCl2, FeCl2, Ca(OH)2, or AL2(SO4)3.
• Biological treatment includes the aerobic and anaerobic treatment of wastewater by a mixed culture of microorganisms.
Certain characteristics of wastewater need to be known before treatment. Among them are physical characteristics, such as color, odor, pH, temperature, and solids contents (suspended and dissolved solids); and chemical characteristics, such as organic and inorganic compounds. Major carbon compounds in a typical industrial waste are carbohydrates, lipids–oils, hydrocarbons, and proteins. Other compounds, such as phenols, surfactants, herbicides, pesticides, and aromatic compounds, are usually in relatively small concentrations (<1 g/l) but are difficult to degrade by biological means. Among inorganic compounds present in wastewater are nitrogenous compounds ![]() , sulfur compounds
, sulfur compounds ![]() , phosphorus compounds
, phosphorus compounds ![]() , heavy metals (Ni2+, Pb2+, Cd2+. Fe2+, Cu2+, Zn2+, Hg2+), and dissolved gases, such as H2S, NH3, and CH4.
, heavy metals (Ni2+, Pb2+, Cd2+. Fe2+, Cu2+, Zn2+, Hg2+), and dissolved gases, such as H2S, NH3, and CH4.
The carbon content (strength) of a wastewater sample can be expressed in several ways: biological oxygen demand (BOD), chemical oxygen demand (COD), and total organic carbon (TOC). Normally, a 5-day BOD value is reported. The BOD5 is the amount of dissolved oxygen (DO) consumed when a wastewater sample is seeded with active bacteria and incubated at 20°C for 5 days. Since the amount of oxygen consumed is stoichiometrically related primarily to the organic content of wastewater, BOD is a measure of the strength of wastewater. This stoichiometric coefficient is not always known, since the composition of the organics is usually unknown. Also, some nitrogen-containing or inorganic compounds will exert an oxygen demand. If the only organic compound is glucose, oxygen consumption can be easily related to the carbon content of wastewater under aerobic conditions:
According to the stoichiometry of this reaction, 1.07 g of oxygen is required for the oxidation of 1 g of glucose.
Samples of wastewater need to be properly diluted to obtain an accurate BOD5 measurement, seeded with active bacteria, and incubated at 20°C for 5 days along with an unseeded blank. BOD5 is calculated using the following equation:
BOD measurements have some shortcomings. This method is applicable only to biodegradable, soluble organics and requires a high concentration of active bacteria preadapted to this type of waste. Moreover, if organic compounds are refractory, 5 days of incubation may not suffice, and 20 days of incubation (BOD20) may be required.
COD is a measure of the concentration of chemically oxidizable organic compounds present in wastewater. Organic compounds are oxidized by a strong chemical oxidant, and using the reaction stoichiometry, the organic content is calculated. Almost all organic compounds present in wastewater are oxidized by certain strong chemical oxidants. Therefore, the COD content of a wastewater sample usually exceeds the measured BOD (COD > BOD5).
A typical chemical oxidation reaction (unbalanced) is
Dichromate may be used as an oxidizing agent, and by a redox balance, the amount of oxygen required to oxidize organic compounds can be calculated. This method is faster (order of 3 hours), easier, and less expensive than BOD measurements.
The TOC content of wastewater samples can be determined by using a TOC analyzer. After proper dilutions, samples are injected into a high-temperature (900° to 950°F) furnace, and all organic carbon compounds are oxidized to CO2, which is measured by an infrared analyzer. To determine the TOC content, wastewater samples should be acidified to remove inorganic carbon compounds (mainly carbonates). The total carbon content of wastewater can be determined before and after acidification, and the difference is inorganic carbon content.
The nitrogen content of wastewater samples is usually measured by total Kjeldahl nitrogen (TKN) determination. Other key nutrient concentrations, such as phosphate, sulfur, and toxic compounds, should be determined before waste streams are treated.
The concentration of biomass in a waste-treatment system using suspended cells is measured as mixed-liquor volatile suspended solids (MLVSS). Basically, wastewater of known volume is filtered, and the collected solids are dried and weighed to give mixed-liquor suspended solids (MLSS). This material is then volatilized by burning in air at 600°C. The weight of the remaining noncombustible, inorganic material is the fixed solids. The difference between the original mass prior to combustion and the fixed solids is the volatile portion. The volatile portion, or MLVSS, is assumed to be primarily microbes, although carbonaceous particles are included in the measurement.
A typical waste-treatment operation employing biological treatment includes the following steps:
1. Primary treatment includes the removal of coarse solids and suspended matter (screening, sedimentation, filtration) and conditioning of the wastewater stream by pH adjustment and nutrient additions (e.g., ![]() ).
).
2. Secondary treatment is the major step in biological treatment; it includes biological oxidation or anaerobic treatment of soluble and insoluble organic compounds. Organic compounds are oxidized to CO2 and H2O by organisms under aerobic conditions. Unoxidized organic compounds and solids from aerobic treatment (e.g., cell wall material, lipids–fats) are decomposed to a mixture of CH4, CO2, and H2S under anaerobic conditions. A sludge of undecomposed material must be purged from either system.
3. Tertiary treatment includes the removal of the remaining inorganic compounds (phosphate, sulfate, ammonium) and other refractory organic compounds by one or more physical separation methods, such as carbon adsorption, deep-bed filtration, and in some cases membrane-based techniques, such as reverse osmosis or electrodialysis.
16.5.1. Biological Waste-Treatment Processes
Biological wastewater treatment usually employs a mixed culture of organisms whose makeup varies with the nature of the waste. Biological treatment may be aerobic or anaerobic. The major aerobic processes (or reactor types) used in wastewater treatment are activated sludge, trickling filter, rotating biological contractors, and oxidation ponds.
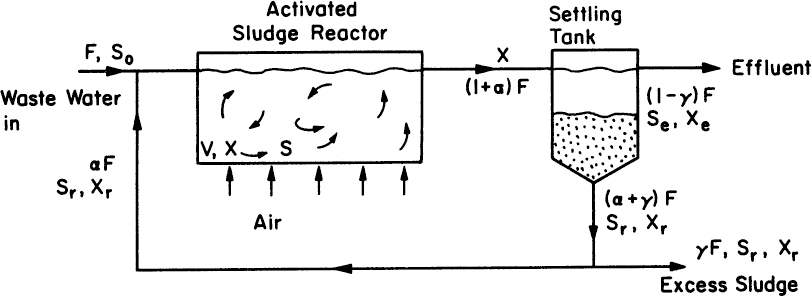
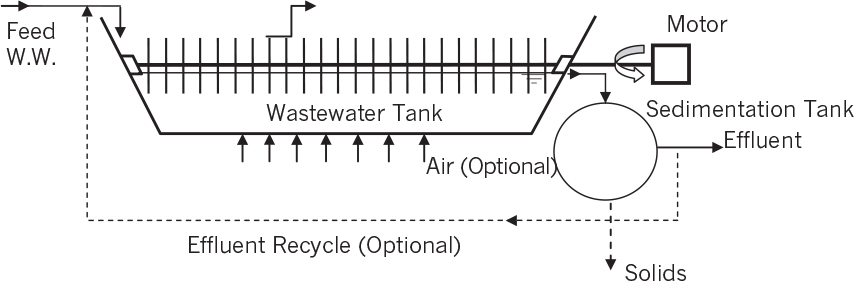
Activated-sludge processes include a well-agitated and aerated continuous-flow reactor and a settling tank. Depending on the physical design of the tank and how feed is introduced into the tank, it may approximate either a plug flow reactor (PFR) or continuous-flow, stirred-tank reactor (CFSTR). A long, narrow tank with single feed approaches PFR behavior; circular tanks approach CFSTR. The concentrated cells from the settling tank are recycled back to the stirred-tank reactor. Figure 16.8 is a schematic of a typical activated-sludge process. Usually, a mixed culture of organisms is utilized in the bioreactor. Some of these organisms may produce polymeric materials (polysaccharides), which help the organisms to agglomerate. Floc formation is a common phenomenon encountered in activated-sludge processes, which may impose some mass-transfer limitations on the biological oxidation of soluble organics; but good floc formation is essential to good performance of the system, since large dense flocs are required in the sedimentation step. Cell recycle from the sedimentation unit improves the volumetric rate of biological oxidation (i.e., high-density culture) and therefore reduces the residence time or volume of the sludge reactor for a given feed rate. The recycle ratio needs to be controlled to maximize BOD removal rate.
The selection of aerator and agitators is a critical factor in the design of activated-sludge processes. The aeration requirements vary depending on the strength of the wastewater and cell concentration. Oxygen requirements for a typical activated-sludge process are about 30 to 60 m3O2/kg of BOD removed. Various aeration devices with and without mechanical agitation can be used in activated-sludge units. Mechanical surface aerators are widely used for shallow activated-sludge units. Surface aerators consist of partially submerged impellers attached to motors mounted on fixed structures. Surface aerators spray liquid and create rapid changes at the air–water interface to enhance oxygen transfer. Pure oxygen may be used for high-strength wastewater treatment. Also, stagewise operation with pure oxygen has been found to be a very effective method of wastewater treatment. The UNOX process, first developed by Union Carbide, is based on this concept. Other forms of aeration include bubble aerators and fixed turbines similar to those we discussed in Chapter 10, “Selection, Scale-Up, Operation, and Control of Bioreactors.”
The activated-sludge system faces many uncontrolled disturbances in input parameters, such as waste flow and composition. Such disturbances can lead to system failure (less-than-adequate treatment of the waste stream). One type of disturbance is shock loading. A shock load indicates the sudden input (pulse) of a high concentration of a toxic compound. A CFSTR design, of course, is less affected by such inputs than a PFR design.
One response to disturbances is sludge bulking. A bulking sludge has flocs that do not settle well, and consequently cell mass is not recycled. Bulking sludge often results from a change in the composition of the microbial population in the treatment unit. For example, filamentous bacteria may dominate the normal floc-forming cells, leading to small, light flocs.
Using rate expressions for microbial growth and substrate utilization and material balances for biomass and substrate, we can determine the required volume of an activated-sludge tank for a certain degree of BOD removal. Since an activated-sludge tank contains a mixed culture of organisms, the actual kinetics of BOD removal are complicated. Usually, interactions among various species are not known. The following analysis is based on pure-culture kinetics, which is only an approximation. It is also assumed that the specific growth-rate expression is given by the Monod equation, with a death rate (or endogenous respiration rate) term:
Following are steady-state material balances for biomass and rate-limiting substrates in an activated-sludge tank (Figure 16.7):
These equations are very similar to the case of the chemostat with recycle discussed in Chapter 9, “Operating Considerations for Bioreactors for Suspension and Immobilized Cultures.” They differ in that we now include the endogenous respiration term.
Assuming no substrate utilization and cell growth in the settling tank (short residence times), material balances around the settling tank have the following yield:
where α is the ratio of sludge recycle flow rate to feed flow rate and is the ratio of excess sludge flow to feed flow rate.
Assuming that the substrate is not separated in the settling tank (that is, S = Se = Sr), equation 16.33 can be eliminated. By rearranging equation 16.32, we can obtain
Substituting equations 16.34 and 16.29 into equation 16.30 yields
Defining μnet = 1/θc, where θc is the cells’ (solids’) residence time, we obtain
Equation 16.36 is used to calculate the cellular (solids’) residence time in the sludge tank. The value of θc is controlled by operator choice of recycle flow rates. Hydraulic (liquid) residence time is
Substituting Sr = S in equation 16.31 gives
or
Equation 16.39 is used to calculate the required volume of the sludge tank for a certain degree of BOD removal (S0– S).
The resulting reactor volume can be expressed in terms of the recycle ratio by substituting equation 16.34 into equation 16.37 to yield
The kinetic parameters of the active organisms in the sludge tank need to be known (i.e., μm, KS, ![]() and kd), and other physical parameters (V, F, α, Xr, X, and S0) need to be determined for the design (sizing, aeration requirement) of activated-sludge units. Typical hydraulic residence time for the aeration tank is 4 to 12 hours and typical sludge age is 3 to 10 days.
and kd), and other physical parameters (V, F, α, Xr, X, and S0) need to be determined for the design (sizing, aeration requirement) of activated-sludge units. Typical hydraulic residence time for the aeration tank is 4 to 12 hours and typical sludge age is 3 to 10 days.
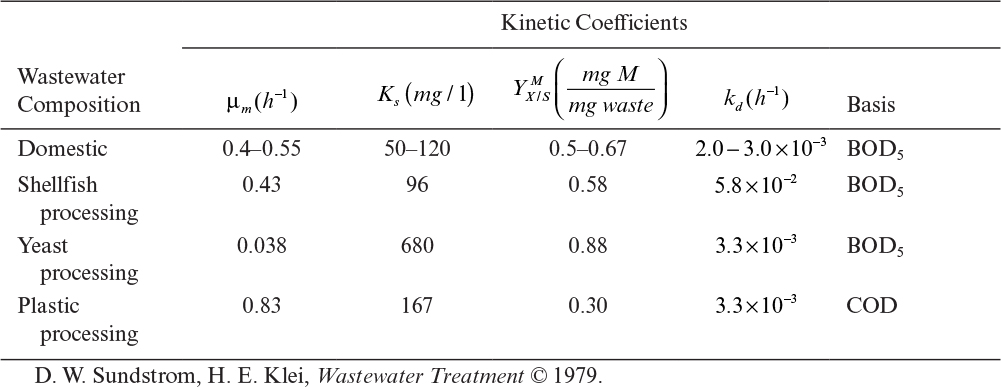
A summary of typical values for the kinetic parameters in biological waste treatment is presented in Table 16.2. Despite all simplifying assumptions, the pure-culture model seems to fit steady-state experimental data reasonably well, although it does not predict the dynamic performance very well.
Although not required by the question, we could use equation 16.40 to get a value of Xr, since α = 0.40. Once Xr is known, γ can be calculated if needed.
Modified activated sludge processes are used in order to improve performance of classical activated sludge processes depending on characteristics of wastewater:
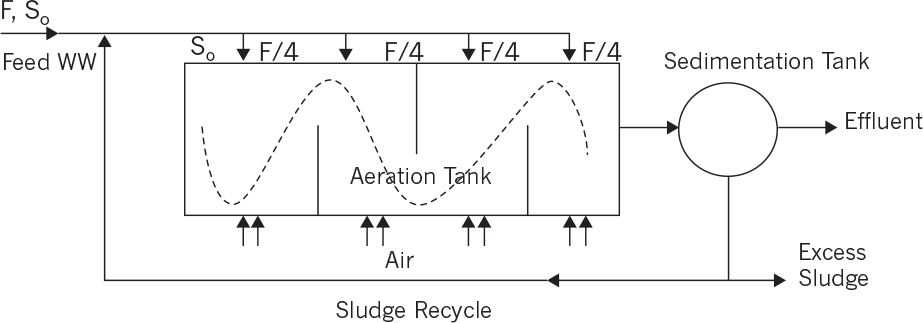
• Step feeding/step aeration is used for high feed flow rates and feed COD concentrations (i.e., high COD loading rates). Long aeration tanks (large L/W ratio) are used, and the wastewater is fed to the reactor at different points along the axis of the aeration tank. Similarly, aeration is provided at different points from the bottom of the tank using finely perforated disks. Figure 16.9 depicts a schematic of the step feeding activated-sludge system. Organic loading is distributed evenly along the length of the aeration tank to avoid shock loadings. The aeration tank behaves like interconnected small tanks in series where the feeding is supplied to each section simultaneously.
• Sludge re-aeration is used when organic compounds are adsorbed on microbial flocs in sedimentation tank and are recycled back to the aeration tank. In order to oxidize adsorbed organics and to keep the thick sludge active, sludge is aerated in the recycle stream by using a small re-aeration tank with a hydraulic residence time (HRT) of less than 1 h. Figure 16.10 depicts a schematic of an activated sludge with sludge re-aeration. This process scheme is also used for wastewaters containing insoluble organics.
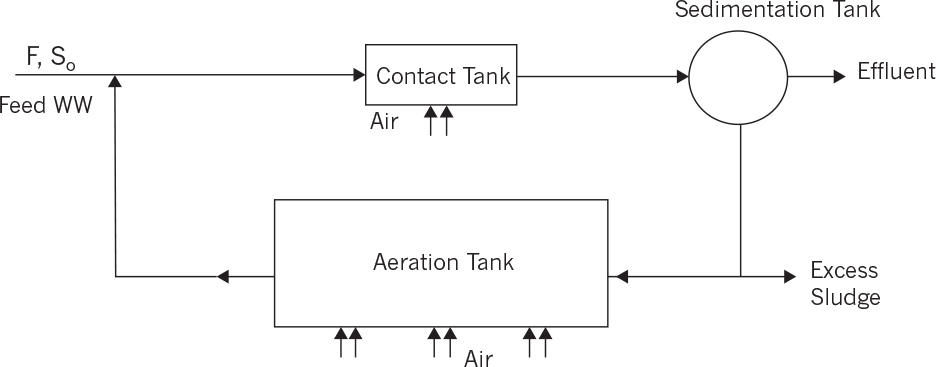
• Contact stabilization process is commonly used for wastewaters containing insoluble organics. Instead of using a primary sedimentation tank and removing insoluble organics, a contact tank is used where the recycled sludge and the feed wastewater are mixed and insoluble organics are entrapped among microbial flocs. Insoluble organics are settled along with microbial flocs in sedimentation tank and are oxidized in an aeration tank in the sludge recycle stream. HRT of the contact tank is less than 1 h, whereas HRT of the aeration tank is between 6 and 10 h depending on the effluent COD. Figure 16.11 depicts a schematic of a contact stabilization process. The major advantage of this process is elimination of primary sedimentation tank and reduction in overall HRT or reactor volumes (space).
• Adsorbent-added activated-sludge processes are used for wastewaters containing refractory (nonbiodegradable) organic compounds. Usually, powdered activated carbon (PAC) is added to the aeration tank or injected to the feed wastewater due to high adsorption capacity of PAC. Refractory organics are adsorbed onto PAC surfaces and are removed from wastewater by the excess sludge. PAC also stimulates floc formation, yielding better sedimentation of sludge and therefore high biomass concentrations in the recycle and also in the aeration tank. Therefore, smaller aeration tanks (low HRT) with lower aeration requirement may be used in this process as compared to the classical activated-sludge systems.
• Oxygen-enriched air or pure oxygen may be used for treatment of high-strength wastewaters containing refractory compounds. Use of oxygen-enriched air or pure oxygen increases solubility and diffusivity of oxygen in the aeration tank, yielding high COD removal rates. The original UNOX process uses multistage, sealed wastewater treatment tanks in series where pure oxygen is provided by using diffusors and is well distributed inside the reactors. High biomass concentrations with high COD removal rates can be achieved by using pure oxygen instead of air. Figure 16.12 depicts a schematic of the UNOX process.
Trickling biological filters consist of a packed bed of inert support particles (sand or plastics) covered with a mixed culture of microorganisms in the form of a film or slime layer and cell aggregates. The column is loosely filled with packing material to yield a void fraction of 0.4 to 0.5. The filter bed is usually arranged in the form of a rather shallow bed with a high diameter-to-height ratio (D/H ≈ 3) to avoid possible clogging problems and axial variations. Waste-liquid is fed to the top of the bed using rotary liquid distributors. The waste-liquid flow rate should be low enough to avoid creating shear forces that would remove biofilms from the surfaces of support particles. Air enters the bed from the bottom and moves upward by natural convection. The driving force for air circulation is the temperature difference created by heat released by biological oxidations. The reaction medium is highly heterogeneous, having temperature, pH, DO, and nutrient profiles throughout the column. The thickness of the biofilm and the composition of the organisms in the biofilm may vary with the length of the column. DO limitations are likely because of the high density of cells, unfavorable hydrodynamic conditions, and diffusion barriers within the film. Pre-aeration of the feed wastewater stream (to saturate with oxygen) and high liquid circulation rates may partly alleviate oxygen transfer problems.
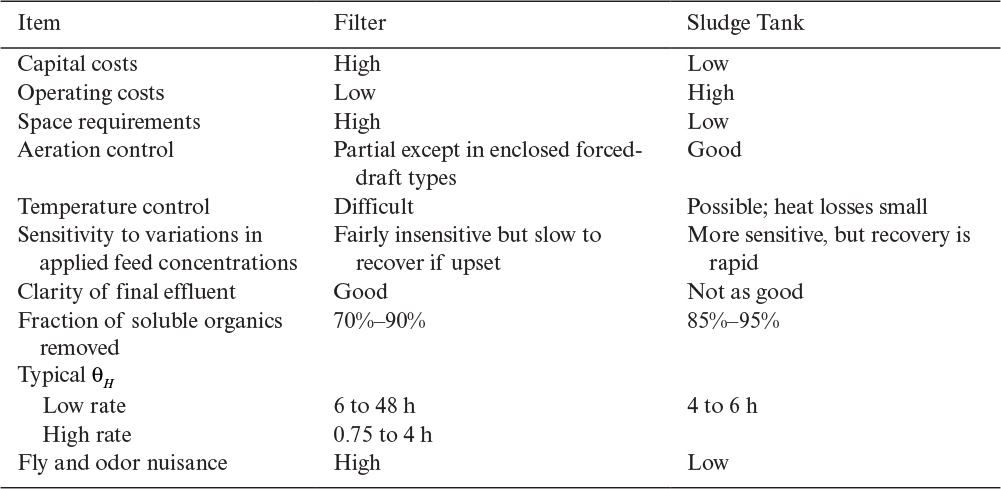
As the liquid flows downward in the column on the surface of microbial films, organic compounds (substrates) diffuse through the microbial film and are utilized by organisms simultaneously. The liquid film over the biomass film should be thin enough to allow adequate aeration. A typical liquid film thickness is on the order of 0.01 mm, and biofilm thickness is 0.25 mm. Typical hydraulic residence times in trickling biological filters (TBF) are 0.5 to 4 h for high-rate filters using recirculation of effluent. Trickling filters are more stable against shock loads than are activated-sludge units. Trickling filters also entail lower operating costs and often give better effluent clarity compared to activated-sludge units. However, capital cost and space requirements for trickling filters are higher than for activated-sludge units, and trickling filters remove a smaller fraction of the soluble organics. Also, the maximum concentration of BOD in the influent is more constrained in trickling filters than in activated-sludge systems. A major problem with the operation of trickling filter units is poor control of conditions (T, pH, DO) due to the heterogeneous nature of the system. A comparison of TBFs with activated-sludge units is presented in Table 16.3.
The liquid effluent of TBFs is usually recycled to obtain more complete removal of BOD from wastewater streams. Figure 16.13 is a schematic of a trickling biological filter.
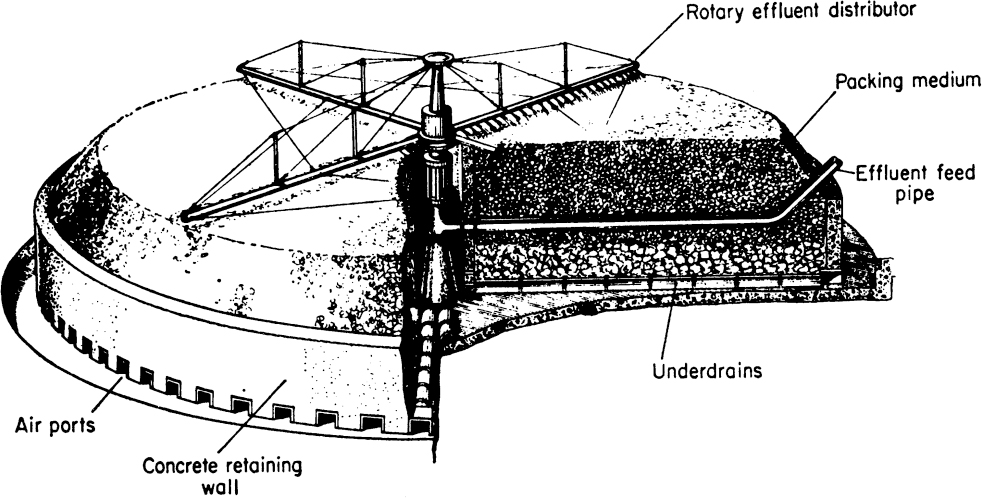
Figure 16.13. Typical trickling biological filter. (With permission, from J. W. Abson and K. H. Todhunter, in N. Blakebrough, ed., Biochemical and Biological Engineering Science, Vol. 1, Academic Press, New York, 1967, p. 326.)
The substrate balance on a differential height of a trickling biological filter yields
where F is liquid flow rate (l/h), Ns is the substrate flux or rate of substrate utilization per unit surface area of biofilm (mg S/cm2 film h), a is biofilm surface area per unit volume of the bed (cm2 film/cm3), and A (cm2) and z (cm) are the cross-sectional area and the height of the bed, respectively.
The substrate flux or rate of substrate consumption is
where η is the effectiveness factor, L is the thickness of the biofilm (cm), rm is the maximum rate of removal of substrate (mg S/l s), and S0 is the substrate concentration in the bulk liquid (mg/cm3). Typical values for rm are 0.2 to 0.5 mg S/l-s. Substituting equation 16.42 into equation 16.41 yields
Equation 16.43 can be integrated to yield a substrate concentration profile throughout the column. Usually, the substrate concentration in bulk liquid is very low, and the biological rate expression can be approximated to first order:
Integration of equation 16.44 from z = 0 to z = H yields
where S0i is the inlet substrate concentration, and S0 is concentration of substrate in the liquid phase at a distance z from the inlet. Experimental data of ln S0/S0i plotted versus z yield a straight line (in at least some cases), supporting the validity of this analysis.
The effectiveness factor can be calculated by using the following expressions:
where
Here, ![]() and rm = μr X/YX/S. De is the effective diffusivity of the substrate within the biofilm (cm2/s), and Yx/s is the yield coefficient (g cells/g substrate).
and rm = μr X/YX/S. De is the effective diffusivity of the substrate within the biofilm (cm2/s), and Yx/s is the yield coefficient (g cells/g substrate).
Following is a similar but more empirical design equation that has been used to determine waste-treatment parameters for biological filter:
where a is the specific surface area of the inert support material (cm2/cm3), A is the cross-sectional area of the bed, F is liquid flow rate, and K is the apparent rate coefficient. In practice, K is used as an adjustable parameter in a curve-fitting procedure. Exponents m and n vary depending on system characteristics such as geometry, hydrodynamics, biological systems, and wastewater characteristics.
A schematic of a typical rotating biological contactor (RBC) is depicted in Figure 16.14, and the disks are shown in Figure 16.15. Disks are made of polystyrene, polyurethane, or PVC with 2 to 4 m diameter and are mounted on a shaft located inside a wastewater tank. The shaft is rotated (10 to 20 rpm) with the aid of a motor. Microorganisms grow in form of biofilm on surfaces of rotating disks. Aeration takes place by direct contact of biofilm with air during rotation. Part of the biofilm surface is immersed in wastewater at any time during rotation where oxidations of organic compounds take place. Biofilm thickness should be controlled in order to avoid thick biofilm formation and anaerobiosis inside the biofilm. For thin biofilms, COD removal may be limited by insufficient biomass in RBC; for thick biofilms, diffusion limitations inside biofilm may cause reductions in COD removal rates. Therefore, the system should be operated around the optimal biofilm thickness to maximize COD removal rate. Control of biofilm thickness is a major problem in this system just as in any other biofilm systems. In RBC systems, COD removal is mainly achieved by the biofilm bacteria. However, in some cases, suspended bacteria may also contribute COD removal depending on relative magnitudes of total biomass in suspended culture and in biofilm.

Figure 16.15. Styrofoam disks used in rotating biological contactor units (Biosystems Division, Autotrol Corporation). (With permission, from E. D. Schroeder, Water and Wastewater Treatment, McGraw-Hill Book Co., New York, 1977, p. 306.)
Assuming a completely mixed aqueous phase in the wastewater tank, COD balance around the RBC tank yields
where F is feed wastewater flow rate (m3 h–1); S0 and S are the feed and effluent COD (kgm–3); Rsf and Rss are the COD removal rates by biofilm and suspended bacteria, respectively; Vf and VL are the biofilm and wastewater volumes (m3) in the tank; L is biofilm thickness (assumed to be constant) and Aw (m2) is the wet biofilm surface area on the disks.
Rsf and Rss can be expressed as
where kf and ks are the maximum COD removal rate constants (μm/YG) by the biofilm and suspended bacteria (kg COD kg–1 biomass h–1); Xf and Xs are the biomass concentrations in biofilm and in wastewater (kg X m–3); Sf and S are the average COD concentrations in biofilm and in wastewater (kg COD m–3); Ksf and Kss are saturation constants for biofilm and suspended bacteria (kg S m–3); and η is the effectiveness factor representing diffusion limitations inside biofilm. For thin biofilms where Sf = S, η = 1; for thick biofilms where Sf << S, η < 1.
Usually, VLXs << Vf Xf and therefore Rss << Rsf. That is, COD removal by suspended cells is usually negligible as compared to that of the biofilm for low-strength wastewaters. However, this assumption may not hold for high-strength wastewaters with liquid-phase aeration.
Therefore, with negligible Rss, equation 16.50 takes the following form:
where Rm (= kf Xf) is the maximum rate of COD removal (kg COD m–3 h–1); Aw is the wet disk surface area.
Aw = α AT where α is the immersion ratio, and AT is the total surface area of disks:
where Ro and Ru are the outer and unwetted radius of disks (m), and N is the number of disks. Immersion ratio is usually 40% to 45%, and therefore Aw = 0.40 AT.
Equation 6.52 is used to determine the required disk surface area for a certain degree of COD removal (i.e., known S0 and S). Xf is usually difficult to determine, and for this reason X = Xf L is defined as surface concentration of biomass (kg X m–2 disk surface), assuming constant biofilm thickness. Then equation 6.52 becomes
where Rms = Rm L. Equation 16.54 can be used to determine the kinetic constants using experimental data obtained at different flow rates or HRTs.
For small RBC tanks with liquid-phase aeration, completely mixed reactor assumption may be valid. However, for long RBC tanks with still water, conditions may approach plug flow regime. In this case, a differential balance over the RBC tank yields
Integration of equation 16.55 results in equation 16.56:
where Af is the biofilm surface area (= Aw, m2) and Xs is the surface biomass concentration (= Xf L, kg biomass m–2 surface area). Equation 16.56 can be used to determine required biofilm and disk surface area for certain degree of COD removal. The effectiveness coefficient (η) is determined by using Figure 9.15 or equations 9.62 to 9.64.
Oxidation ponds provide another inexpensive alternative to activated-sludge and TBF operations. Oxidation ponds are shallow (2 to 4 ft deep) waste-treatment reactors closely resembling natural aquatic ecosystems. Bacteria and algae grow in the same pond in a symbiotic relationship. Bacteria oxidize organic compounds by utilizing oxygen produced by algae and produce CO2; algae utilize CO2 produced by bacteria and produce oxygen by photosynthesis for bacterial consumption. Such ponds require large land areas, are less efficient than many other techniques, and may have adverse environmental side effects. Toxic or hazardous materials may collect in the sediment without degradation, creating a long-term problem.
Anaerobic digestion (or biological treatment) is usually used to treat solid wastes and excess sludge produced in aerobic waste-treatment processes. Particulate waste material removed by screening or sedimentation in primary waste treatment and biomass (concentrated sludge) produced in activated-sludge units are degraded under anaerobic conditions to produce methane. Anaerobic digestion is a slow process compared to aerobic processes; typical residence times are 30 to 60 days. The microbiology and biochemistry of anaerobic digestion are very complicated. However, the major steps involved in this process are as follows:
1. Solubilization of insoluble organics: Waste material may contain large amounts of solids made of cellulosics (papers, agricultural wastes), starches (potato waste), and other complex insoluble organic chemicals. Solubilization of these compounds by acid or enzymatic hydrolysis (cellulases, amylases, glucoamylases, lipases, proteases) is the first step in anaerobic digestion. These compounds are not readily utilizable by microorganisms, and their hydrolysis is essential for effective microbial digestion.
2. Formation of volatile acids: Solubilized organic compounds are metabolized by anaerobic bacteria to produce volatile organic acids, such as acetic, butyric, formic, and propionic acids, and short-chain fatty acids. Acid-producing organisms are a mixture of facultative anaerobes, such as enteric bacteria and clostridial species, which are called acid formers. Alcohol formation (butanol, propanol, ethanol) also takes place to a lesser extent. The optimal temperature and pH values for this step are T = 35°C and pH = 4 to 6. The partial pressure of H2 in the reactor can greatly influence metabolism.
3. Formation of methane: Volatile acids and alcohols produced in the second stage are converted to methane and CO2 by methanogenic bacteria, which are strictly anaerobic. Among methanogenic bacteria used for this purpose are Methanobacterium (nonspore-forming rods), Methanobacillus (spore-forming rods), and Methanococcus and Methanosarcina (a cocci group growing in cubes of eight cells). The optimal temperature and pH range for methanogenic bacteria are T = 35° to 40°C and pH = 7 to 7.8.
The major biological reaction steps involved in a typical anaerobic digestion process can be represented as follows:
Depending on the composition of organic waste, either enzymatic hydrolysis of insolubles (e.g., cellulosics) or methane production from volatile acids is the rate-limiting step in this reaction sequence. The concentration of H2 is often critical in determining the rates of these processes. Hydrolysis reactions are usually carried out as a separate step. However, acid-formation and methane-formation steps can be achieved in the same reactor. Single- and two-reactor systems have been developed for anaerobic digestion. The two-step reactor scheme has been reported to result in higher reaction rates and methane yields. The operating conditions in a single-reactor system are pH = 6 to 7 and T = 35° to 40°C, which result in reasonable rates of acid formation and methane generation. Typical solids residence time in an anaerobic digester is 10 to 30 days.
Cell yields in anaerobic digesters are low, and a typical value is 0.05 g cells/g COD. Cell yield also varies with substrate. Yield on carbohydrates is larger than yield on the other carbon compounds.
Changes in temperature affect the methane-formation rate more than the acid-formation rate. Although the optimal temperature is reported to be 35° to 40°C, in some cases higher removal rates are obtained at higher temperatures (T = 55°C), depending on the composition of the organisms. The system does not function satisfactorily at temperatures below 35°C, and the temperature of the reactors needs to be controlled for stable methane generation.
The rate of methane formation is considerably lower than the rate of acid formation and is rate limiting. Sudden increases in organic load concentrations may cause volatile acid (VA) accumulation in the medium, decreasing pH and altering H2 levels, which further depresses methane production. Typical soluble organics removal rates are on the order of 0.2 g COD/g cells day. Certain metal ions (Na+, K+, Ca2+, Mg2+) are known to be toxic to organisms of anaerobic digesters at high concentrations (>1000 mg/l), although they are stimulatory at low levels (>100 mg/l).
The composition of the product gas mixture varies, depending on the composition of waste material and other environmental factors. A typical gas composition is 70% to 75% CH4, 20% to 25% CO2, and 5% H2S and other gases (NO2, H2, CO). Part of the methane produced in an anaerobic digestion unit is used to heat the digesters to fermentation temperature. The digester gas has a heating value of 24 to 28 million J/m3, and the yield is 0.75 to 1.2 m3 (std)/kg of organic material decomposed, resulting in an energy generation of 18 to 33 million J/kg of organic material. One problem associated with the use of digester gas is the H2S content, which can cause foul odors and corrosion and needs to be removed before use.
Anaerobic digesters were originally designed as closed stationary tanks. In newer designs, reactor contents are mixed and the reactor is heated. Mixing is accomplished either by recycling a portion of the gas or by mechanical stirring. The reaction mixture is highly heterogeneous and viscous and therefore difficult to mix. Figure 16.16 is a schematic diagram of an anaerobic digester. Anaerobic digestion can be achieved in packed beds containing porous solid support particles. Bacteria grow on support surfaces and inside pores, and liquid waste may be trickled through the bed. Solids recycling schemes have been developed for use with conventional digesters that simulate activated-sludge units. Solids residence times are considerably higher than hydraulic residence times in these systems, which results in biomass accumulation and higher reaction rates. Continuous PFRs operated in upflow mode are commonly used as anaerobic digesters.
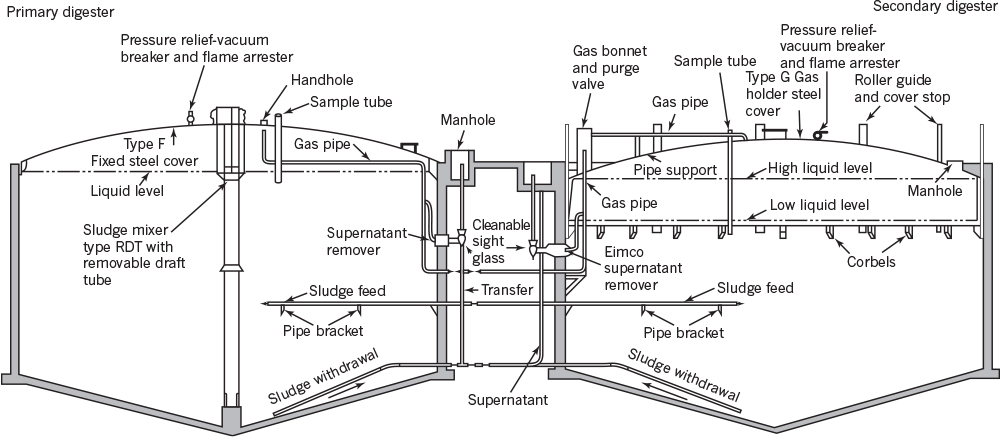
Figure 16.16. A schematic of an anaerobic digestion unit (Eimco Processing Machinery Division, Envirotech Corporation). (With permission, from E. D. Schroeder, Water and Wastewater Treatment, McGraw-Hill Book Co., New York, 1979, p. 327.)
The solids content of sludge (waste) is reduced by about 50% to 60% at the end of anaerobic digestion. Digested sludge is much less biodegradable than raw sludge and is easier to dewater. The digester effluent is dewatered by vacuum filtration and is dried. The dried solids are either incinerated or spread on land as fertilizer.
16.5.2. Advanced Wastewater Treatment Systems
Advanced wastewater treatment systems are used for removal of residual nitrogen and phosphorus along with refractory carbonaceous compounds. Examples are for nitrification and denitrification, phosphate removal processes (e.g., PhoStrip and A/O processes), and combined nitrogen and phosphate removal processes (e.g., A2O and Bardenpho processes).
Nitrogen-containing organic compounds (e.g., amino acids and proteins) are oxidized biologically to ammonium, which is further oxidized to nitrite and nitrate by Nitrosomonas and Nitrobacter, respectively. The conversion of ammonium to nitrate by microbial catalysis is called nitrification:
Overall, combining equations 16.57a and 16.57b results in equation 16.58:
Both of the aforementioned organisms fix carbon dioxide to be used for biosynthesis. Aerobic nitrification can take place during BOD removal in the presence of these organisms and increases the oxygen requirements in aerobic treatment.
These reactions are energy yielding, and the first step (equation 16.57a) is the rate-limiting one. Nitrification processes can be accomplished in a single stage with BOD removal or as a two-stage system with separate BOD removal and nitrification units. The choice between the one-stage and two-stage processes typically depends on the ratio of carbon to nitrogen in the feed stream. Typically, this ratio is expressed as BOD5/TKN. The growth yield of nitrifying bacteria is low (0.2 g dry wt per g N) and the fraction of the population that is nitrifying bacteria can be low. For example, at BOD5/TKN = 10, the fraction of nitrifying bacteria is 0.02; at BOD5/TKN = 0.5, the fraction of nitrifying bacteria rises to 0.35. For a single-stage unit with combined BOD removal and nitrification, the BOD5/TKN ratio should be greater than 5. When it is less than 3, the two-stage process is preferred.
Nitrification is best accomplished with suspended cells, typically using a modification of the activated-sludge process. However, fixed-film processes that are well aerated can yield nitrification efficiencies over 90%. In either system, DO levels should be high (>2mg/l), and slightly alkaline solutions (7.5 to 8.5) at 20° to 30°C work best.
Under anaerobic conditions, some bacteria utilize nitrate as the final electron acceptor instead of oxygen. This process is known as denitrification. Two types of nitrate reduction are possible: assimilatory and dissimilatory reduction. In assimilatory reduction, nitrate is reduced to ammonia and is incorporated into cell biomass. However, in dissimilatory reduction, nitrate is reduced to nitrite and further to elemental nitrogen. An external carbon source is needed; this source must be inexpensive (e.g., waste starch, methanol, or molasses).
An example of assimilatory reduction follows:
And following is an example of dissimilatory reduction:
Using an average biomass composition of C5H7NO2 and assuming that methanol is used as the carbon source, the overall balance for denitrification can be written as follows:
Clearly, equations such as 16.62 can be used to predict the amount of carbon source (e.g., methanol) required to remove a known amount of ![]() . A number of microbes can act as denitrifiers; among these are species of the genera Pseudomonas, Alcaligenes, Arthrobacter, and Corynebacter. Such bacteria grow slowly with μg = 0.5 d–1 and with a yield coefficient of 0.8 g biomass per g nitrogen removed. Saturation constants are low (e.g., 0.1 mg/l < K5 < 1 mg/l) for both nitrification and denitrification, so these reactions can be approximated as zero order for nitrogen concentrations above about 2 mg/l.
. A number of microbes can act as denitrifiers; among these are species of the genera Pseudomonas, Alcaligenes, Arthrobacter, and Corynebacter. Such bacteria grow slowly with μg = 0.5 d–1 and with a yield coefficient of 0.8 g biomass per g nitrogen removed. Saturation constants are low (e.g., 0.1 mg/l < K5 < 1 mg/l) for both nitrification and denitrification, so these reactions can be approximated as zero order for nitrogen concentrations above about 2 mg/l.
Activated-sludge or packed-bed biofilm type reactors may be used for anaerobic denitrification. Upflow packed-bed reactors utilizing biofilms on porous support material are reported to result in high nitrate removal rates.
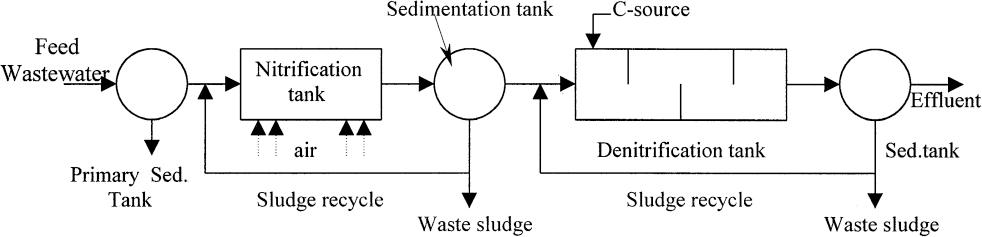
A typical two-stage nitrification/denitrification process is depicted in Figure 16.17. Optimal pH and temperature values for denitrification are pH = 6.5 to 7 and T = 20° to 30°C.
Phosphate removal from wastewaters by biological means can be realized either by assimilation or by “luxury” phosphate uptake. Assimilation of phosphate occurs in all aerobic/anaerobic processes, since nearly 3% of cell mass is made of phosphorus. Luxury phosphate uptake is accomplished by organisms belonging to the genera Acinetobacter, among which Acinetobacter calcoaceticus is the most widely known. Acinetobacter species utilize carbohydrates as carbon and energy source by the Entner–Doudoroff pathway. Those organisms utilize acetate and fatty acids under anaerobic conditions to synthesize polyhydroxybutyrate (PHB). Energy required for PHB synthesis is obtained from ATP molecules generated by breakdown of polyphosphates. Under aerobic conditions in the absence of carbon source, the organisms utilize PHB molecules as carbon–energy source to synthesize polyphosphates and store them in form of polyphosphate granules inside the cell.
An example of aerobic polyphosphate synthesis follows:
The following is an example of anaerobic polyphosphate depolymerization:
Consequently, the organisms remove phosphate from liquid media to synthesize and store polyphosphates under aerobic conditions and release phosphate into the media under anaerobic/anoxic conditions. Acinetobacter species can store polyphosphates up to 30% of their dry weight.
In phosphate removal bioprocesses, organisms are exposed to alternating anaerobic and aerobic conditions. The two major processes developed to promote luxury phosphate uptake are the A/O (anaerobic/oxic) and PhoStrip processes.
The A/O process is used for combined BOD and phosphate removal. It is a two-stage process incorporating anaerobic and oxic (aerobic) steps in sequence. In the anaerobic stage, the phosphate in the recycled sludge is released into liquid media. The released phosphate is taken up by the cells under aerobic conditions along with phosphate in the feed wastewater stream and is stored as polyphosphate granules. Polyphosphate is removed from the system by the waste sludge, which has a high value as fertilizer. When the BOD/P ratio in the feed wastewater is greater then 10, the effluent phosphate levels can be lower than 1 mg/l (as shown in Figure 16.17).
The PhoStrip process consists of an aerobic stage and an anaerobic one on the sludge recycle stream, as shown in Figure 16.18. The wastewater stream is fed to the aerobic stage for polyphosphate synthesis, the effluent of which is fed to a sedimentation tank. The underflow of the sedimentation tank is fed to the anaerobic stage for phosphate release. The released phosphate is precipitated in a sedimentation tank with the addition of lime in form of calcium phosphate. Part of the sedimented sludge and anaerobic reactor sludge is recycled back to the aerobic stage. Phosphate is removed from the process in the form of calcium phosphate. The effluent water stream would contain less then 1.5 mg/l phosphate in a well-operating PhoStrip process.
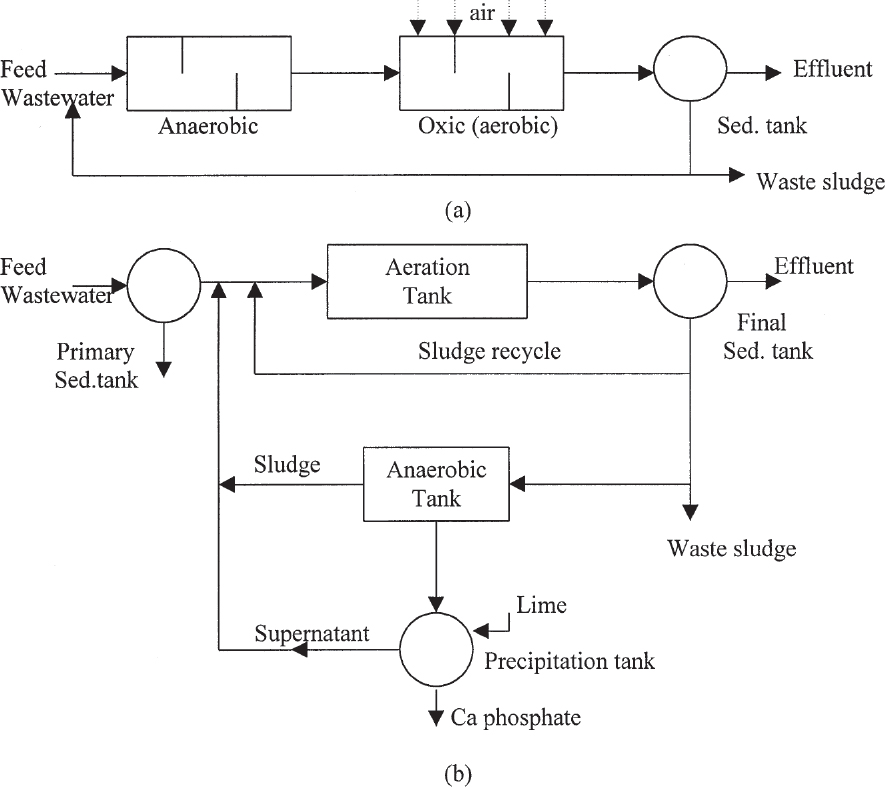
Combined nitrogen and phosphate removal processes employ combinations of anaerobic, anoxic, and aerobic zones in different orders which accomplish simultaneous BOD removal also. The most commonly used processes for this purpose are the A2/O and the five-stage Bardenpho processes (see Figure 16.19). Among other processes are the University of Capetown (UCT) and the Virginia Initiative Plant (VIP) processes.
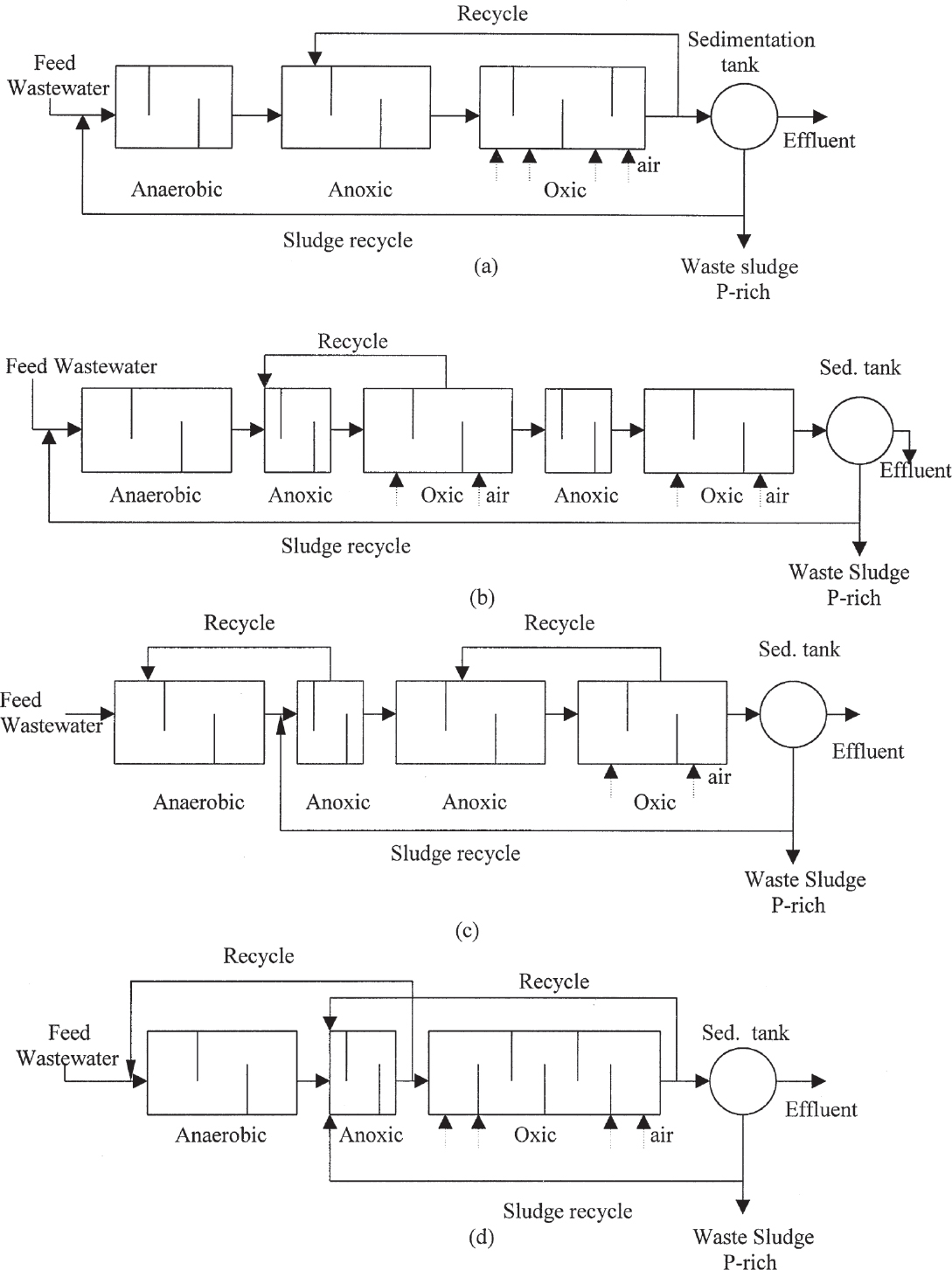
Figure 16.19. Combined biological nitrogen and phosphate removal processes: (a) A2/O process, (b) five-stage Bardenpho process, (c) University of Capetown process, (d) Virginia Initiative Plant process.
The A2/O process includes an anoxic zone for denitrification in addition to A/O process (as shown in Figure 16.19a). Hydraulic residence times in anaerobic and anoxic zones are less than 1.5 h, and in the aerobic zone 4 to 6 h. The effluent of the aerobic stage is recycled back to the anoxic zone for denitrification purposes. Phosphate release takes place in the anaerobic zone. Denitrification and BOD removal are the major functions of the anoxic zone. Phosphate removal in the form of polyphosphates, nitrification, and some BOD removal take place in aerobic zone. Phosphate is removed from the system in form of waste sludge; nitrogen is released in form of N2 (gas); and BOD is converted to CO2 and H2O.
The five-stage Bardenpho process includes two additional anoxic and aerobic zones as compared to the A2/O process (Figure 16.19b). This process is known as the A2/O/A/O process. The effluent of the aerobic zone is partly recycled back to the anoxic zone for denitrification purposes. The last two zones are for additional denitrification and polyphosphate removal to further reduce nitrate and phosphate levels in the effluent. Hydraulic residence times in anaerobic and anoxic zones are less than 4 h, and in the first aerobic zone, 4 to 12 h.
The UCT and VIP processes employ similar zones and recycle schemes in different orders, resulting in low nitrogen, phosphate, and BOD levels in the effluent.
In some cases, sulfur removal from wastewater is needed. Elemental sulfur in wastewater can be oxidized to sulfate by Thiobacillus species. Sulfate can be further reduced to sulfides by anaerobic sulfate-reducing bacteria, such as Desulfovibrio, and sulfides can be precipitated out of wastewater in the presence of certain metal ions, such as Fe2+, Zn2+, and Pb2+. Sulfates can also be precipitated with the addition of limestone (CaCO3) and Ca(OH)2 and can be filtered out of wastewater.
16.5.3. Conversion of Wastewater to Useful Products
Some waste materials containing starch and cellulose or other easily utilizable carbonaceous compounds can easily be converted to useful products such as high-protein feedstuff or SCP, ethanol, organic acids (acetic, butyric, propionic), methane, and methanol. Agricultural wastes, waste paper, and waste wood constitute major cellulosic wastes. Major starch-containing wastes include some agricultural wastes (wheat, corn, rice, potato), some domestic solid wastes (vegetable/fruit), and some industrial wastes (food industry).
Cellulosic and starch-containing wastes are first hydrolyzed into sugar molecules, which are further converted to SCP under aerobic conditions or ethanol/organic acids under anaerobic conditions. While many organisms can be used, yeasts, particularly Saccharomyces, are well suited to protein production, since they already have FDA approval for use in foods. The waste must be free of toxic contaminants. To insure complete utilization of carbon, mineral nutrients may need to be added to give a C/N/P ratio of 100/15/3.
Two widely known processes for conversion of wastes to SCP are the SYMBA and the PEKILO processes. The SYMBA process converts potato wastes to SCP and consists of two stages. The first stage is for enzymatic hydrolysis of starch by Endomycopsis sp., and the second is for SCP production on glucose by Saccharomyces sp. The PEKILO process is based on growth of Paecilomyces sp. on waste sulfite liquor.
Such waste materials can also be converted to chemicals such as ethanol for use as a fuel, solvents such as acetone/butanol, or monomers for synthesis of polymers (lactic acid). Conversion of starches to glucose and fermentable sugars is a well-established industry that uses large quantities of enzymes such as α-amylase, β-amylase, and glucoamylase. The economically feasible conversion of cellulosic wastes into fermentable sugars remains a bioprocess challenge. Many types of cellulases made by microbes can be used; new processes using genetically engineered microbes (e.g., Trichoderma sp., Thermomonospora sp., and some Clostridia sp.) may lead to less expensive cellulases. Acid hydrolysis can be used instead of enzymatic processes but is less environmentally friendly. The economical conversion of cellulosic wastes into products, such as ethanol, would be a major contribution to human welfare.
16.6. Summary
Populations containing multiple species are important in natural ecosystems, well-defined processes, wastewater treatment, and systems using genetically modified cells. Some examples of interactions among these species are competition, neutralism, mutualism, protocooperation, commensalism, amensalism, predation, and parasitism. In real systems, several modes of interaction may be present. Mathematical analyses can be used to show that neither pure competition nor pure mutualism gives a stable steady state in a chemostat. However, spatial heterogeneity, dynamic fluctuations, and the addition of other interactions can lead to the sustained coexistence of species with competitive or mutualistic interactions.
One of the major process uses of mixed cultures is wastewater treatment. The activated-sludge system is commonly employed in treating wastewaters. Such a system can be considered a chemostat with cell recycle under aerobic conditions. Alternative methods of aerobic treatment include trickling-bed filters, rotating biological disks, and oxidation ponds. Anaerobic digestion can be used to directly treat wastewaters or, more commonly, to further degrade sludges from the primary and secondary treatment levels of a waste-treatment plant.
Suggestions for Further Reading
FREDRICKSON, A. G., Behavior of Mixed Cultures of Microorganisms, Ann. Rev. Microbiol. 31: 63–87, 1977.
HAMMER, M. J., AND M. J. HAMMER, JR., Water and Wastewater Technology, 7th ed., Prentice Hall, Upper Saddle River, NJ, 2011.
SCHROEDER, E. D., Water and Wastewater Treatment, McGraw-Hill, New York, 1977.
SUNDSTROM, D. W., AND H. E. KLEI, Wastewater Treatment, Prentice Hall, Englewood Cliffs, NJ, 1979.
TCHOBANOGLOUS, G., F. BURTON, H. D. STENSEL, AND R. TSUCHIHASHI, Wastewater Engineering: Treatment and Resource Recovery (Metcalf & Eddy, Inc.), 5th ed., McGraw-Hill, New York, 2013.
VIESSMAN, W., AND M. J. HAMMER, Water Supply and Pollution Control, 6th ed., Prentice Hall, Upper Saddle River, NJ, 1998.
Problems
16.1. A batch fermenter receives 1 l of medium with 5 g/l of glucose, which is the growth-rate-limiting nutrient for a mixed population of two bacteria (a strain of E. coli and Azotobacter vinelandii). A. vinelandii is five times larger than E. coli. The replication rates for the two organisms are
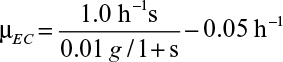
and

The yield coefficients are
YEC = 0.5 gDW/g glucose
YAV = 0.35 gDW/g glucose
The inoculum for the fermenter is 0.03 gDW/l of E. coli (1 × 108 cells/ml) and 0.15 gDW/l of A. vinelandii (1 × 108 cells/ml).
What will be the ratio of A. vinelandii to E. coli at the time when all of the glucose is consumed?
16.2. Consider Example 16.1, where we demonstrated that two bacteria competing for a single nutrient in a chemostat (well-mixed) could not coexist. Consider the situation where B can adhere to a surface but A cannot. Redo the balance equations, where a is the surface area available per unit reactor volume and the rate of attachment is first order in XB with a rate constant kaB. The sites available for attachment will be (XAtBM – XAtB)aV. The attached cells can detach with a first-order dependence on the attached cell concentration (XAtB) with a rate constant of kaB. Attached cells grow with the same kinetics as suspended cells.
a. Without mathematical proofs, do you think coexistence may be possible? Why or why not?
b. Consider the following specific case and solve the appropriate balance equations for D = 0.4 h–1:
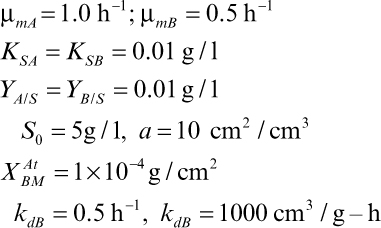
16.3. Organism A grows on substrate S and produces product P, which is the only substrate that organism B can utilize. The batch kinetics are as follows:
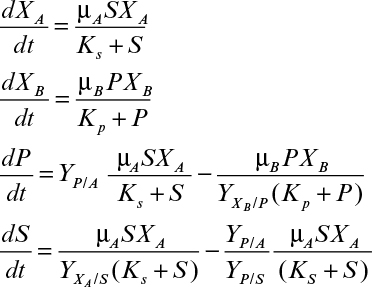
Assume the following parameter values:

Determine the behavior of these two organisms in a chemostat. Plot S, P, XA, and XB versus dilution rate. Discuss what happens to organism B as the dilution rate approaches the washout dilution rate for organism A.
(Courtesy of L. Erickson, from “Collected Coursework Problems in Biochemical Engineering,” compiled by H. W. Blanch for 1977 American Society for Engineering Education Summer School.)
16.4. The BOD5 value of a wastewater feed stream to an activated-sludge unit is S0 = 300 mg/l, and the effluent is desired to be S = 30 mg/l. The feed flow rate is F = 2 × 107 l/day. For the recycle ratio of α = 0.5 and a steady-state biomass concentration of X = 5 g/l, calculate the following:
a. Required reactor volume (V)
b. Biomass concentration in recycle (Xr)
c. Solids (cells) residence time (θc)
d. Hydraulic residence time (θH)
e. Daily oxygen requirement
Use the following kinetic parameters:

16.5. For the activated-sludge unit shown in Figure 16.8, the specific growth rate of cells is given by the following equation:
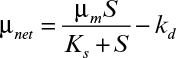
The following parameter values are known: F = 500 l/h, α = 0.4, γ = 0.1, Xe = 0, V = 1500 l, Ks = 10 mg/l, μm = 1 h–1, kd = 0.05 h–1, S0 = 1000 mg/l, ![]() = 0.5 gDW/g substrate.
= 0.5 gDW/g substrate.
a. Calculate the substrate concentration (S) in the reactor at steady state.
b. Calculate the cell concentration(s) in the reactor.
c. Calculate Xr and Sr in the recycle stream.
16.6. Wastewater with a flow rate of 6 m3/h and COD0 (S0) = 1000 mg/l is treated in an activated sludge unit with aeration tank volume of 50 m3 to obtain CODe = 30 mg/l in the effluent. The following information are available:
k = 5 d–1 = μm/Y, Ks = 125 mg/l, Y = 0.35 gX/gS, b= kd = 0.03 d–1, Xr = 10 g/l
a. Determine θc (SRT), θH (HRT), X and the sludge recycle ratio (r).
b. If the sludge recycle ratio is r = 0.3, what should be the flow rate of wastewater in order to obtain CODe = 30 mg/l. Determine X and θH for this case.
16.7. Wastewater with a flow rate of 10 m3/h and COD0 = 2000 mg/l is treated in an activated-sludge unit. The following information are available:
k = 6 d–1, Ks = 150 mg/l, Y = 0.4 gX/gS, b = kd = 0.02 d–1, Xr = 10 g/l, r = 0.4 Determine θcm, θc, CODe (S), θH, V, X, and sludge production rate if the aeration tank is completely mixed and θc = 4θcm.
16.8. An aerated lagoon is an activated sludge system without sludge recycle with only a completely mixed and continuously operating aeration tank. If the wastewater flow rate is 5 m3/h, COD0 = 500 mg/l and the effluent COD is desired to be 30 mg/l in an aerated lagoon.
a. Determine θH (HRT), θc (SRT), biomass concentration (X), and the volume of the aeration tank (V) by using first-order kinetics.
b. Determine θH, θc, biomass concentration (X) and the volume of the aeration tank (V) by using the Monod kinetics.
c. Compare and comment on the results.
The kinetic constants are k = 4 d–1, Ks = 100 mg/l, Y = 0.35 gX/gS, and b = kd = 0.
16.9. An activated-sludge waste treatment system is required to reduce the amount of BOD5 from 1000 mg/l to 20 mg/l at the exit. The sedimentation unit concentrates biomass by a factor of 3. Kinetic parameters are μm = 0.2 h–1, Ks = 80 mg/l, kd = 0.01 h–1, and ![]() = 0.5 g MLVSS/g BOD5. The flow of wastewater is 10,000 l/h, and the size of the treatment basin is 50,000 l.
= 0.5 g MLVSS/g BOD5. The flow of wastewater is 10,000 l/h, and the size of the treatment basin is 50,000 l.
a. What is the value of the solids residence time (i.e., θc)?
b. What value of the recycle ratio must be used?
16.10. Consider a well-mixed waste-treatment system for a small-scale system. The system is operated with a reactor of 1000 l and flow rate of 100 l/h. The separator concentrates biomass by a factor of 2. The recycle ratio is 0.7. The kinetic parameters are μm = 0.5 h–1, Ks = 0.2 g/l, Y MX/S = 0.5 g/g, and kd = 0.05 h–1. What is the exit substrate concentration?
16.11. Redo Example 16.4 if the Contois equation for growth applies. In this case, use the following equation:
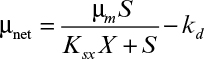
The values of μm and kd are the same as for Example 16.4, but Ks no longer applies. Assume Ksx = 0.02 g BOD5/g MLVSS.
16.12. In a trickling biological filter, the BOD value of the feed stream is S0i = 500 mg/l with a feed flow of F = 103 l/h. The effluent BOD value is desired to be S0 = 10 mg/l. The following kinetic parameters for the biocatalysts are known: rm = 20 mg/S/l-h and Ks = 200 mgS/l. The biofilm thickness is L = 0.1 mm. The cross-sectional area of the filter is A = 2 m2, and the biofilm surface area per unit volume of the bed is a = 500 cm2/cm3. Assume that dissolved oxygen is the rate-limiting substrate and the diffusion coefficient of oxygen is DO2 = 2 × 10–5 cm2/s. Determine the required height of the bed. You can assume first-order bioreaction kinetics.
16.13. Wastewater with a flow rate of Q = 15 m3/h and COD0 = 1500 mg/l is treated in a trickling biological filter with effluent recycle to obtain CODe = 50 mg/l in the effluent. The following data are available:
Y = 0.35 gX/gS, μm = 0.1 h–1, X = 15 g/l, Ks = 150 mg/l, L = 1.5 mm, DO/H = 4, H = 2.5 m
a. Determine the effluent recycle ratio (R) in order to obtain Si = 500 mg/l in the feed wastewater.
b. Determine the biofilm surface area (af and Af) and particle surface area (ap and Ap) by assuming only 65% of the particle surfaces are covered by biofilm.
c. When af = 100 m2/m3, what would be the DO and H, if DO/H = 4, for the same performance of the TBF?
Use generalized design equation for TBF:
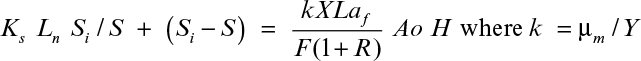
16.14. Wastewater with COD0 = 800 mg/L is treated by using a single-stage RBC to obtain a treatment efficiency of E = 1 – S/S0 = 0.95. The following information is available:
k = 4 d–1, X = 100 g/m2, Ks= 120 mg/L, DO = 3 m, N = 300, immersion ratio is 0.40
a. What should be the flow rate of wastewater in order to obtain 95% COD removal if RBC tank is completely mixed? Determine the wet and total surface area of the disks (Aw, AT) for this case.
b. Determine the flow rate of wastewater (F), Aw, and AT, if the RBC tank behaves like a plug flow reactor.
c. Compare AT/F values for both cases and comment on the results. Which reactor is better and why?
16.15. Wastewater with a flow rate of Q = 15 m3/h and COD0 = 1500 mg/l is treated by using a two stage RBC unit connected in series. The effluent of the second stage is desired to be S2 = 50 mg/l. COD removal efficiencies for each stage are equal, E1 = E2. The following information is available:
k = 3 d–1, X1 = 90 g/m2, X2 = 75 g/m2, Ks = 100 mg/l, DO = 4 m, immersion is 40%
a. Determine S1, Aw1, Aw2, AWT, AT1, AT2, and AT.
b. Determine the number of disks for each stage, N1, N2, and NT.