 B. Ideal Gas Constant and Conversion Factors
by H. Scott Fogler
Elements of Chemical Reaction Engineering, Fifth Edition
B. Ideal Gas Constant and Conversion Factors
by H. Scott Fogler
Elements of Chemical Reaction Engineering, Fifth Edition
- About This E-Book
- Title Page
- Copyright Page
- Dedication Page
- Contents
- Preface
- A. Who Is the Intended Audience?
- B. What Are the Goals of This Book?
- C. What Is the Structure of CRE?
- D. What Are the Components of the CRE Web Site?
- E. Why Do We Assign Homework Problems?
- F. What Is a Living Example Problem (LEP)?
- G. What Software Is Available to Solve the LEPs?
- H. Are There Other Web Site Resources?
- I. How Can Critical Thinking and Creative Thinking Skills Be Enhanced?
- I.2 Enhance Creative Thinking Skills
- J. What’s New in This Edition?
- J.2 Content
- K. How Do I Say Thank You?
- About the Author
- 1. Mole Balances
- 2. Conversion and Reactor Sizing
- 3. Rate Laws
- 4. Stoichiometry
- 5. Isothermal Reactor Design: Conversion
- 5.1 Design Structure for Isothermal Reactors
- 5.2 Batch Reactors (BRs)
- 5.3 Continuous-Stirred Tank Reactors (CSTRs)
- 5.4 Tubular Reactors
- 5.5 Pressure Drop in Reactors
- 5.6 Synthesizing the Design of a Chemical Plant
- Summary
- ODE Solver Algorithm
- CRE Web Site Materials
- Questions and Problems
- Supplementary Reading
- 6. Isothermal Reactor Design: Moles and Molar Flow Rates
- 6.1 The Molar Flow Rate Balance Algorithm
- 6.2 Mole Balances on CSTRs, PFRs, PBRs, and Batch Reactors
- 6.3 Application of the PFR Molar Flow Rate Algorithm to a Microreactor
- 6.4 Membrane Reactors
- 6.5 Unsteady-State Operation of Stirred Reactors
- 6.6 Semibatch Reactors
- Summary
- ODE Solver Algorithm
- CRE Web Site Materials
- Questions and Problems
- Supplementary Reading
- 7. Collection and Analysis of Rate Data
- 7.1 The Algorithm for Data Analysis
- 7.2 Determining the Reaction Order for Each of Two Reactants Using the Method of Excess
- 7.3 Integral Method
- 7.4 Differential Method of Analysis
- 7.5 Nonlinear Regression
- 7.6 Reaction-Rate Data from Differential Reactors
- 7.7 Experimental Planning
- Summary
- CRE Web Site Materials
- Questions and Problems
- Supplementary Reading
- 8. Multiple Reactions
- 8.1 Definitions
- 8.2 Algorithm for Multiple Reactions
- 8.3 Parallel Reactions
- 8.4 Reactions in Series
- 8.5 Complex Reactions
- 8.6 Membrane Reactors to Improve Selectivity in Multiple Reactions
- 8.7 Sorting It All Out
- 8.8 The Fun Part
- Summary
- CRE Web Site Materials
- Questions and Problems
- Supplementary Reading
- 9. Reaction Mechanisms, Pathways, Bioreactions, and Bioreactors
- 10. Catalysis and Catalytic Reactors
- 10.1 Catalysts
- 10.2 Steps in a Catalytic Reaction
- 10.3 Synthesizing a Rate Law, Mechanism, and Rate-Limiting Step
- 10.3.1 Is the Adsorption of Cumene Rate-Limiting?
- 10.3.2 Is the Surface Reaction Rate-Limiting?
- 10.3.3 Is the Desorption of Benzene Rate-Limiting?
- 10.3.4 Summary of the Cumene Decomposition
- 10.3.5 Reforming Catalysts
- 10.3.6 Rate Laws Derived from the Pseudo-Steady-State Hypothesis (PSSH)
- 10.3.7 Temperature Dependence of the Rate Law
- 10.4 Heterogeneous Data Analysis for Reactor Design
- 10.5 Reaction Engineering in Microelectronic Fabrication
- 10.6 Model Discrimination
- 10.7 Catalyst Deactivation
- Summary
- CRE Web Site Materials
- Questions and Problems
- Supplementary Reading
- 11. Nonisothermal Reactor Design—The Steady-State Energy Balance and Adiabatic PFR Applications
- 12. Steady-State Nonisothermal Reactor Design—Flow Reactors with Heat Exchange
- 12.1 Steady-State Tubular Reactor with Heat Exchange
- 12.2 Balance on the Heat-Transfer Fluid
- 12.3 Algorithm for PFR/PBR Design with Heat Effects
- 12.4 CSTR with Heat Effects
- 12.5 Multiple Steady States (MSS)
- 12.6 Nonisothermal Multiple Chemical Reactions
- 12.7 Radial and Axial Variations in a Tubular Reactor
- 12.8 Safety
- Summary
- CRE Web Site Materials
- Questions and Problems
- Supplementary Reading
- 13. Unsteady-State Nonisothermal Reactor Design
- 14. Mass Transfer Limitations in Reacting Systems
- 15. Diffusion and Reaction
- 15.1 Diffusion and Reactions in Homogeneous Systems
- 15.2 Diffusion and Reactions in Spherical Catalyst Pellets
- 15.3 The Internal Effectiveness Factor
- 15.4 Falsified Kinetics
- 15.5 Overall Effectiveness Factor
- 15.6 Estimation of Diffusion- and Reaction-Limited Regimes
- 15.7 Mass Transfer and Reaction in a Packed Bed
- 15.8 Determination of Limiting Situations from Reaction-Rate Data
- 15.9 Multiphase Reactors in the Professional Reference Shelf
- 15.10 Fluidized Bed Reactors
- 15.11 Chemical Vapor Deposition (CVD)
- Summary
- CRE Web Site Materials
- Questions and Problems
- Journal Critique Problems
- Supplementary Reading
- 16. Residence Time Distributions of Chemical Reactors
- 17. Predicting Conversion Directly from the Residence Time Distribution
- 18. Models for Nonideal Reactors
- 18.1 Some Guidelines for Developing Models
- 18.2 The Tanks-in-Series (T-I-S) One-Parameter Model
- 18.3 Dispersion One-Parameter Model
- 18.4 Flow, Reaction, and Dispersion
- 18.5 Tanks-in-Series Model versus Dispersion Model
- 18.6 Numerical Solutions to Flows with Dispersion and Reaction
- 18.7 Two-Parameter Models—Modeling Real Reactors with Combinations of Ideal Reactors
- 18.8 Use of Software Packages to Determine the Model Parameters
- 18.9 Other Models of Nonideal Reactors Using CSTRs and PFRs
- 18.10 Applications to Pharmacokinetic Modeling
- Summary
- CRE Web Site Materials
- Questions and problems
- Supplementary Reading
- A. Numerical Techniques
- B. Ideal Gas Constant and Conversion Factors
- C. Thermodynamic Relationships Involving the Equilibrium Constant
- D. Software Packages
- E. Rate-Law Data
- F. Nomenclature
- G. Open-Ended Problems
- G.1 Design of Reaction Engineering Experiment
- G.2 Effective Lubricant Design
- G.3 Peach Bottom Nuclear Reactor
- G.4 Underground Wet Oxidation
- G.5 Hydrodesulfurization Reactor Design
- G.6 Continuous Bioprocessing
- G.7 Methanol Synthesis
- G.8 Cajun Seafood Gumbo
- G.9 Alcohol Metabolism
- G.10 Methanol Poisoning
- H. Use of Computational Chemistry Software Packages
- I. How to Use the CRE Web Resources
- Index
B. Ideal Gas Constant and Conversion Factors
Ideal Gas Constant

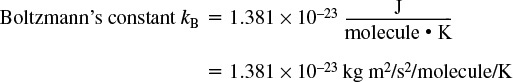
Volume of Ideal Gas
1 lb-mol of an ideal gas at 32°F and 1 atm occupies 359 ft3 (0.00279 lbmol/ft3).
1 mol of an ideal gas at 0°C and 1 atm occupies 22.4 dm3 (0.0446 mol/dm3).
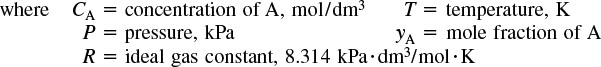
Viscosity
1 poise = 1 g/cm/s = 0.1 kg/m/s
1 centipoise = 1 cp = 0.01 poise = 0.1 micro Pascal · second
Force
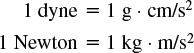
Pressure
1 Pa = 1 Newton/m2
Work
A. Work = Force × Distance
1 Joule = 1 Newton · meter =1 kg m2/s2 = 1 Pa · m3
B. Pressure × Volume = Work
(1 Newton/m2) · m3 = 1 Newton · m = 1 Joule
Time Rate of Change of Energy with Time
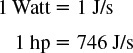
Gravitational Conversion Factor
Gravitational constant
g = 32.2 ft/s2
American Engineering System
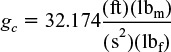
SI/cgs System
gc = 1 (Dimensionless)
-
No Comment
..................Content has been hidden....................
You can't read the all page of ebook, please click here login for view all page.


