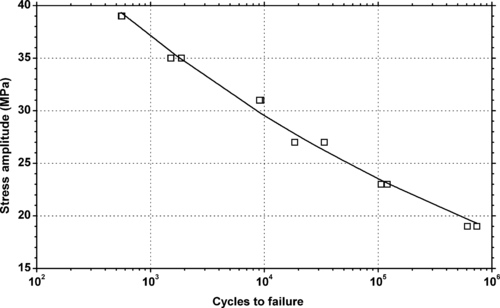
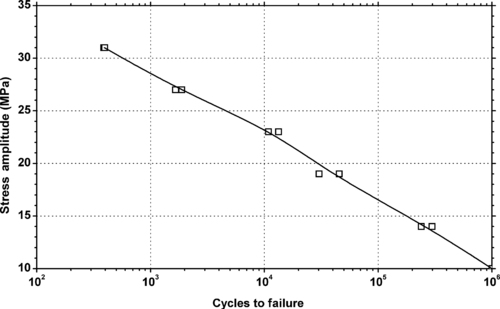
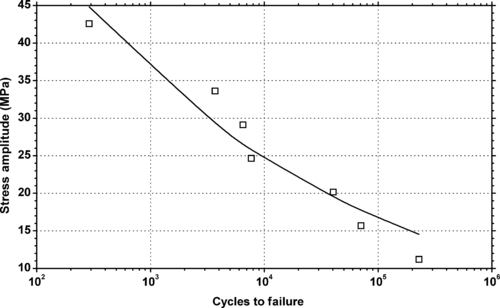
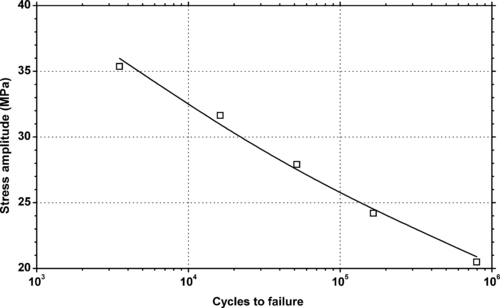
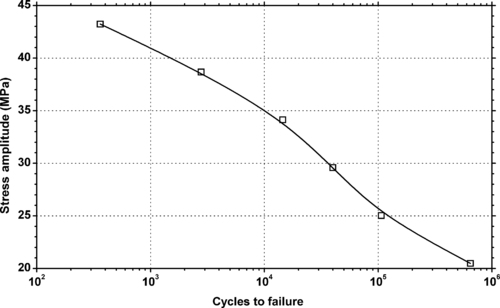
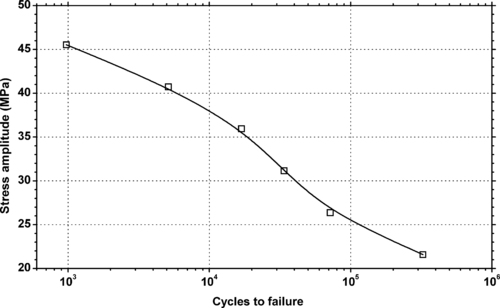
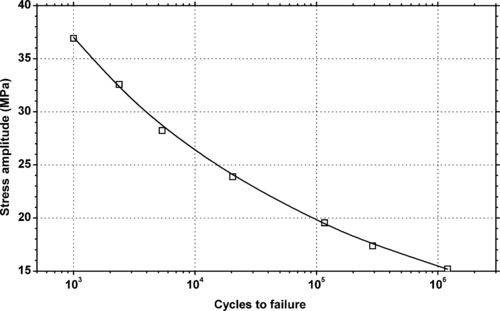
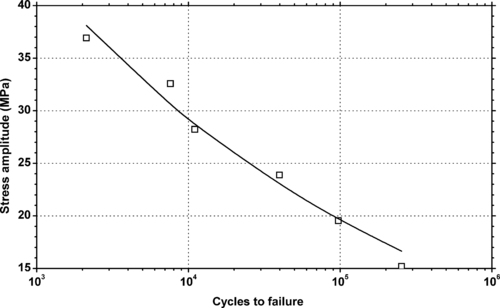
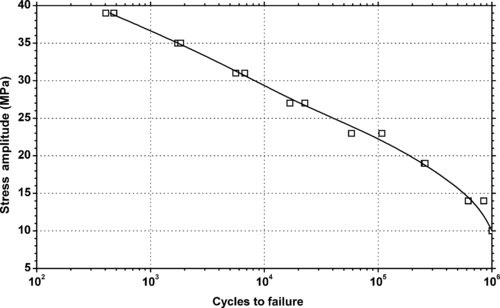
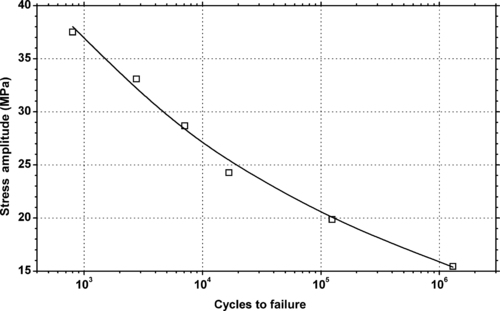
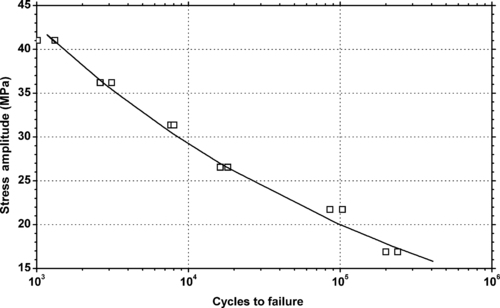
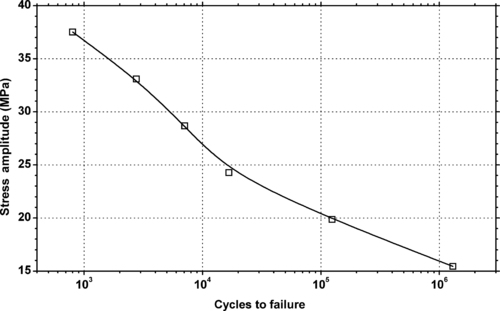
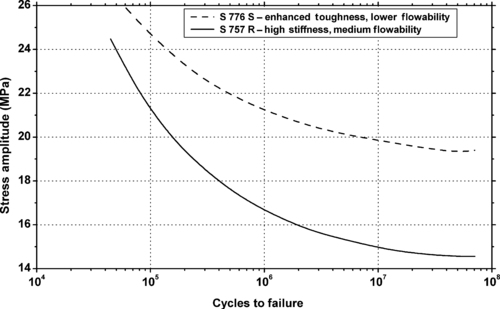
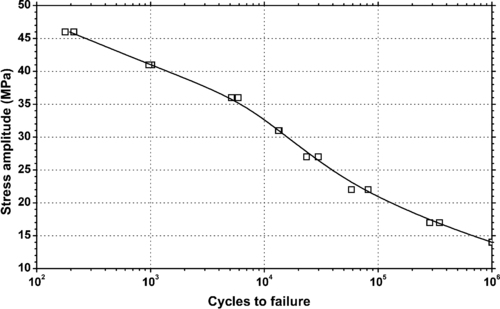
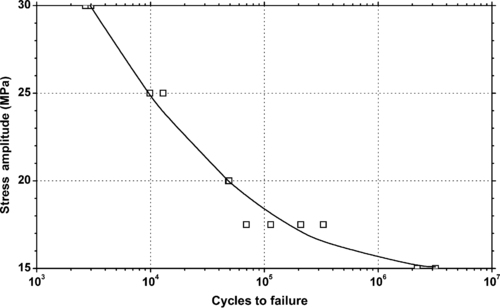
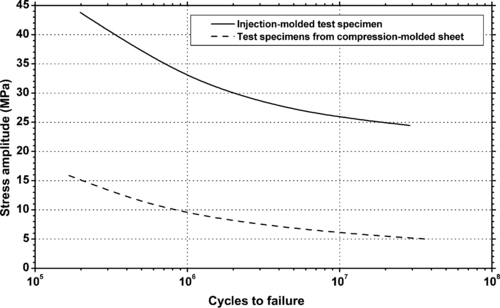
4.1. Background
This chapter on styrenic plastics covers a broad class of polymeric materials of which an important part is styrene. Styrene, also known as vinyl benzene, is an organic compound with the chemical formula C
6H
5CH = CH
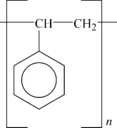
2. Its structure is shown in
Figure 4.1.
It is used as a monomer to make plastics such as polystyrene, acrylonitrile butadiene styrene (ABS), styrene acrylonitrile (SAN), and the other polymers in this chapter.
4.1.1. Polystyrene
Polystyrene is the simplest plastic based on styrene. Its structure is shown in
Figure 4.2.
Pure solid polystyrene is a colorless, hard plastic with limited flexibility. Polystyrene can be transparent or can be made in various colors. It is economical and is used for producing plastic model assembly kits, plastic cutlery, CD “jewel” cases, and many other objects where a fairly rigid, economical plastic is desired.
Polystyrene’s most common use, however, is expanded polystyrene (EPS). EPS is produced from a mixture of about 5–10% gaseous blowing agent (most commonly pentane or carbon dioxide) and 90–95% polystyrene by weight. The solid plastic beads are expanded into foam through the use of heat (usually steam). The heating is carried out in a large vessel holding 200–2000 liters. An agitator is used to keep the beads from fusing together. The expanded beads are lighter than unexpanded beads so they are forced to the top of the vessel and removed. This expansion process lowers the density of the beads to 3% of their original value and yields a smooth-skinned, closed cell structure. Next, the preexpanded beads are usually “aged” for at least 24 hours in mesh storage silos. This allows air to diffuse into the beads, cooling them, and making them harder. These expanded beads are excellent for detailed molding. Extruded polystyrene (XPS), which is different from EPS, is commonly known by the trade name Styrofoam™. All these foams are not of interest in this book.
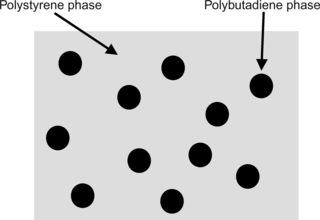
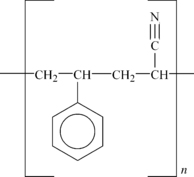
One of the most important plastics is high-impact polystyrene, or HIPS. This is a polystyrene matrix that is imbedded with an impact modifier, which is basically a rubber like polymer such as polybutadiene. This is shown in
Figure 4.3.
4.1.2. Acrylonitrile Styrene Acrylate
ASA is the acronym for acrylate rubber–modified styrene–acrylonitrile copolymer. ASA is a terpolymer that can be produced by either a reaction process of all three monomers or a graft process. ASA is usually made
by introducing a grafted acrylic ester elastomer during the copolymerization of styrene and acrylonitrile, known as SAN. SAN is described in the next section of this chapter. The finely divided elastomer powder is uniformly distributed and grafted to the SAN molecular chains. The outstanding weatherability of ASA is due to the acrylic ester elastomer. ASA polymers are amorphous plastics, which have mechanical properties similar to those of the ABS resins described in
Section 4.1.4. However, the ASA properties are far less affected by outdoor weathering.
ASA resins are available in natural, off-white, and a broad range of standard and custom-matched colors. ASA resins can be compounded with other polymers to make alloys and compounds that benefit from ASA’s weather resistance. ASA is used in many products including lawn and garden equipment, sporting goods, automotive exterior parts, safety helmets, and building materials.
4.1.3. Styrene Acrylonitrile
Styrene and acrylonitrile monomers can be copolymerized to form a random, amorphous copolymer that has good weatherability, stress crack resistance, and barrier properties. The copolymer is called styrene acrylonitrile or SAN. The SAN copolymer generally contains 70–80% styrene and 20–30% acrylonitrile. It is a simple random copolymer. This monomer combination provides higher strength, rigidity, and chemical resistance than polystyrene, but it is not quite as clear as crystal polystyrene and its appearance tends to discolor more quickly. The general structure is shown in
Figure 4.4.
SAN is used for household goods and tableware, in cosmetics packaging, sanitary and toiletry articles as well as for writing materials and office supplies.
4.1.4. Acrylonitrile Butadiene Styrene
Acrylonitrile butadiene styrene, or ABS, is a common thermoplastic used to make light, rigid, molded products such as pipe, automotive body parts, wheel covers, enclosures, and protective head gear.
SAN copolymers have been available since the 1940s and while its increased toughness over styrene made it suitable for many applications, its limitations led to the introduction of a rubber, butadiene, as a third monomer producing the range of materials popularly referred to as ABS plastics. These became available in the 1950s and the availability of these plastics and ease of processing led ABS to become one of the most popular of the engineering polymers.
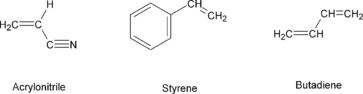
The chemical structures of the monomers are shown in
Figure 4.5. The proportions of the monomers typically range from 15% to 35% acrylonitrile, 5% to 30% butadiene and 40% to 60% styrene. It can be found as a graft copolymer, in which SAN polymer is formed in a polymerization system in the presence of polybutadiene rubber latex; the final product is a complex mixture consisting of SAN copolymer, a graft polymer of styrene acrylonitrile and polybutadiene and some free polybutadiene rubber.
4.1.5. Methyl Methacrylate Acrylonitrile Butadiene Styrene
Methyl methacrylate acrylonitrile butadiene styrene, or MABS, is a newer modification of ABS. It is sometimes called transparent ABS, a copolymer of methyl methacrylate, acrylonitrile, butadiene, and styrene (MABS). Key properties of MABS are excellent transparency, high-impact strength, and good chemical resistance. This is an exceptional combination of properties for an impact-modified thermoplastic. MABS can be used to create particularly brilliant visual effects such as very deep colors, pearly or sparkle effects. It is easy to process and can also be printed upon.
4.1.6. Styrene Maleic Anhydride
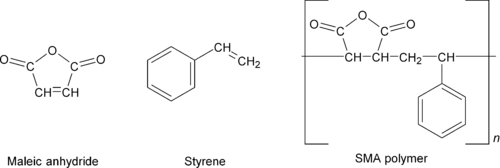
Copolymerization of styrene with maleic anhydride creates a copolymer called styrene maleic anhydride (SMA). This reaction is shown in
Figure 4.6. SMA has a higher glass transition temperature than polystyrene and is chemically reactive because of active functional groups. Thus, SMA polymers are often used in blends or composites where interaction or reaction of the maleic anhydride provides for desirable interfacial effects. The anhydride reaction with primary amines is particularly potent.
4.1.7. Styrenic Block Copolymers
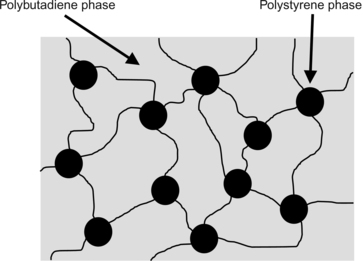
Styrenic block copolymer, or SBC, is a commercially important thermoplastic elastomer. The polymer is made of three separate polymeric blocks (see
Section 3.2 for an explanation of block copolymers). At one end is a hard polystyrene block, in the middle a long polybutadiene (or other elastomeric) block, followed by a second hard block of polystyrene. These blocks are immiscible, so they form discrete domains of polystyrene within a polybutadiene matrix. The separate domains are chemically connected. This is shown in
Figure 4.7, where one might notice that this looks a lot like HIPS, except that the continuous phase and hard discrete phase are switched in SBC and the domains are connected. One additional property of interest is that some SBCs blend well with general-purpose polystyrene, allowing customization of properties.
4.1.8. Styrenic Blends
While the number of styrenic blends might seem limitless, compatibility and morphology limit blend types. Styrenic blends are numerous but most are limited to only a couple of types. The most important blend is ABS and polycarbonate (PC). Next in importance is ABS and polyamide (or nylon, PA). Polystyrene and polyethylene are often used in expandable foams. Polystyrene and polyphenylene ether (PPE or PPO) are commercially important blends, which are covered in a later chapter. The other classes of the styrenic blends are not major product lines but can be very important in some applications.