- Cover
- Title Page
- Contents
- Forword
- Preface
- About the Authors
- 1. Water Technology
- 1.1 Introduction
- 1.2 Sources of Water
- 1.3 Types of Impurities Present in Water
- 1.4 Hard Water and Hardness
- 1.5 Determination of Hardness
- 1.6 Dissolved Oxygen (DO)
- 1.7 Determination of Chlorides in Water
- 1.8 Determination of Acidity in Water
- 1.9 Alkalinity of Water
- 1.10 Disadvantages of Hard Water
- 1.11 Quality of Water for Domestic Use
- 1.12 Treatment of Water for Domestic Use
- 1.13 Break-Point Chlorination
- 1.14 Boilers and Boiler Troubles
- 1.15 Softening of Water
- 1.16 Desalination
- 1.17 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short Answer Questions
- Descriptive Questions
- Problems for Practice
- 2. Polymers
- 2.1 Introduction
- 2.2 Degree of Polymerisation
- 2.3 Classification of Polymers
- 2.3.1 Classification Based on Source
- 2.3.2 Classification Based on Composition
- 2.3.3 Classification Based on Chemical Composition
- 2.3.4 Classification Based on Structure
- 2.3.5 Classification Based on Mode of Polymerisation
- 2.3.6 Classification Based on the Molecular Forces
- 2.3.7 Classification Based on Tacticity
- 2.4 Types of Polymerization
- 2.5 Molecular Mass of a Polymer
- 2.6 Plastics
- 2.7 Important Polymers—Composition, Preparation, Properties and Engineering Uses
- 2.8 Rubber (Elastomers)
- 2.9 Reinforced or Filled Plastics
- 2.10 Biopolymers
- 2.11 Conducting Polymers
- 2.12 Polyphosphazenes/Phosphonitrilic Polymers
- 2.13 Composites
- 2.14 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short Answer Questions
- Descriptive Questions
- 3. Fuels and Combustion
- 3.1 Introduction
- 3.2 Classification of Fuels
- 3.3 Units of Heat
- 3.4 Calorific Value
- 3.5 Determination of Calorific Value
- 3.6 Characteristics of Good Fuel
- 3.7 Solid Fuels
- 3.8 Liquid Fuels
- 3.9 Synthetic Petrol
- 3.10 Power Alcohol
- 3.11 Knocking
- 3.12 Diesel Engine, Cetane and Octane Number
- 3.13 Gaseous Fuels
- 3.14 Flue Gas Analysis by Orsats Apparatus
- 3.15 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short Answer Questions
- Descriptive Questions
- 4. Alternate Energy Resources
- 5. Electrochemistry and Batteries
- 5.1 Introduction
- 5.2 Electrolysis
- 5.3 Electrolytic Conduction
- 5.3.1 Factors Affecting Electrolytic Conduction
- 5.3.2 Electrical Resistance and Conductance
- 5.3.3 Specific, Equivalent and Molar Conductivities
- 5.3.4 Equivalent Conductivity
- 5.3.5 Molar Conductivity
- 5.3.6 Measurement of Electrolytic Conductance
- 5.3.7 Variation of Conductivity with Concentration
- 5.3.8 Conductance Behaviour of Strong Electrolyte
- 5.3.9 Conductance Behaviour of Weak Electrolyte
- 5.4 Kohlrausch’s Law of Independent Migration of Ions
- 5.5 Conductometric Titrations
- 5.6 Electrochemical Cells
- 5.7 Types of Electrodes
- 5.8 Reference Electrode
- 5.9 Ion Selective Electrodes (ISE)
- 5.10 Glass Electrode
- 5.11 Concentration Cell
- 5.12 Potentiometric Titrations
- 5.13 Electrochemical Sensors
- 5.14 Voltammetry
- 5.15 Batteries
- 5.16 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short Answer Questions
- Solved Numerical Problems
- Descriptive Questions
- Problems for Practice
- 6. Science of Corrosion
- 7. Chemistry of Engineering Materials
- 7.1 Semiconducting and Super Conducting Materials
- 7.2 Magnetic Materials
- 7.3 Cement
- 7.3.1 Classification of Cement
- 7.3.2 Raw Materials used in the Manufacture of Portland Cement
- 7.3.3 Manufacture of Portland Cement
- 7.3.4 Chemical Composition of Portland Cement and its Importance
- 7.3.5 Setting and Hardening of Cement
- 7.3.6 ISI Specifications of Cement
- 7.3.7 Analysis of Cement
- 7.3.8 Plaster of Paris/Gypsum Plaster
- 7.4 Refractories
- 7.5 Lubricants
- 7.5.1 Important Functions of Lubricants
- 7.5.2 Mechanism of Lubrication
- 7.5.3 Classification of Lubricants
- 7.5.4 Properties of Lubricants
- 7.5.5 Redwood Viscometer
- 7.5.6 Engler’s Viscometer
- 7.5.7 Saybolt Viscometer
- 7.5.8 U-Tube Viscometer
- 7.5.9 Conversion of Redwood, Engler and Saybolt Viscosities into Absolute Units
- 7.6 Explosives and Propellants
- 7.6.1 Some Important Terms about Explosives
- 7.6.2 Classification of Explosives
- 7.6.3 Precautions during Storage of Explosives
- 7.6.4 Blasting Fuses
- 7.6.5 Important Explosives and their Preparation
- 7.6.6 Rocket Propellants
- 7.6.7 Characteristics of a Good Propellant
- 7.6.8 Classifications of Propellants
- 7.7 Nanomaterials
- 7.8 Liquid Crystals
- 7.9 Abrasives
- 7.10 Review Questions
- Fill in the Blanks
- Multiple-choice questions
- Short Answer Questions
- Descriptive Questions
- 8. Phase Rule
- 8.1 Introduction
- 8.2 Explanation of the Terms Involved in Phase Equilibria
- 8.3 Phase Rule
- 8.4 Phase Diagrams
- 8.5 One Component System
- 8.6 Two Component System
- 8.7 Heat Treatment of Steel
- 8.8 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short Answer Questions
- Solved Numerical Problems
- Descriptive Questions
- 9. Photochemistry
- 9.1 Introduction
- 9.2 Light Source in Photochemistry
- 9.3 Laws of Photochemistry
- 9.4 Photophysical and Chemical Processes
- 9.5 Quantum Yield and Quantum Efficiency
- 9.6 Photosensitisation
- 9.7 Photodynamic Therapy
- 9.8 Important Photochemical Reactions
- 9.9 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short Answer Questions
- Descriptive Questions
- 10. Surface Chemistry
- 10.1 Introduction
- 10.2 Adsorption
- 10.2.1 Mechanism of Adsorption
- 10.2.2 Adsorption is Exothermic
- 10.2.3 Difference between Adsorption and Absorption
- 10.2.4 Examples of Adsorption, Absorption, and Sorption
- 10.2.5 Positive and Negative Adsorptions
- 10.2.6 Classification of Adsorption
- 10.2.7 Factors Affecting the Adsorption of Gases by Solids
- 10.2.8 Adsorption Isotherms
- 10.2.9 Applications of Adsorption
- 10.3 Colloidal State
- 10.4 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short Answer Questions
- Long Answer Questions
- 11. Thermodynamics
- 11.1 Introduction
- 11.2 Types of Thermodynamic Systems
- 11.3 Intensive and Extensive Properties
- 11.4 Reversible and Irreversible Process
- 11.4.1 Reversible Process
- 11.4.2 Irreversible Process
- 11.4.3 Thermodynamic Processes
- 11.4.4 Isothermal Process or Isothermal Change
- 11.4.5 Indicator Diagram
- 11.4.6 Work Done by a System in an Adiabatic Process
- 11.4.7 First Law of Thermodynamics and its Application
- 11.4.8 Second Law of Thermodynamics
- 11.4.9 Carnot’s Engine, Efficiency
- 11.4.10 Working of Carnot’s Engine
- 11.4.11 Absolute Zero
- 11.4.12 Numerical Problems Based on Carnot’s Cycle
- 11.4.13 Solved Numerical Problems Based on Isothermal and Adiabatic Process
- 11.5 Thermodynamic Potentials and Maxwell Equations
- 11.5.1 Thermodynamic Potential
- 11.5.2 Internal Energy (U)
- 11.5.3 Total Heat Function (H)
- 11.5.4 Helmholtz Function (F)
- 11.5.5 Gibb’s Free Energy or Gibb’s Function (G)
- 11.5.6 Maxwell’s Equations
- 11.5.7 Clausius–Clapeyron Equation
- 11.5.8 Derivation of the Stefan–Boltzmann Law using Maxwell’s Equations
- 11.5.9 Joule–Thomson Effect or Joule–Kelvin Effect
- 11.6 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short Answer Questions
- Descriptive Questions
- 12. Metals in Biological System
- 13. Organometallic Compounds
- 14. Coordination Chemistry
- 14.1 Introduction
- 14.2 Basic Requirements to Formation of Coordination Compound
- 14.3 Nomenclature of Metal Complexes
- 14.4 Theories of Coordination Chemistry
- 14.5 Factors Affecting the Stability of Coordination Compounds
- 14.6 Determination of Complex Ion Formation
- 14.7 Stability of Coordination Compounds
- 14.8 Applications of Coordination Compounds
- 14.9 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short Answer Questions
- Descriptive Questions
- 15. Structure and Reactivity of Organic and Inorganic Molecules
- 15.1 Introduction
- 15.2 Hybridisation
- 15.3 Bond Polarisation
- 15.4 Reaction Intermediates
- 15.5 Molecular Orbital Theory
- 15.5.1 Important Points on Molecular Orbital Diagrams
- 15.5.2 Fundamental Steps for Constructing Molecular Orbitals
- 15.5.3 Five Basic Rules of Molecular Orbital Theory
- 15.5.4 Linear Combination of Atomic Orbitals and Type of Atomic Orbitals
- 15.5.5 Molecular Orbital Energy Level Diagrams of Homo Atomic Molecules
- 15.5.6 Molecular Energy Level Diagrams of Hetero Atomic Molecules
- 15.6 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short Answer Questions
- Descriptive Questions
- 16. Stereochemistry
- 17. Spectroscopy
- 17.1 Introduction
- 17.2 Ultra Violet and Visible Spectroscopy
- 17.2.1 Principle
- 17.2.2 Instrumentation
- 17.2.3 Instrumental Design
- 17.2.4 Electronic Transitions
- 17.2.5 Chromophores
- 17.2.6 Auxochrome
- 17.2.7 Woodward–Fieser Rules
- 17.2.8 Factors Affecting the Position of the λ Maximum and Intensity of Radiation
- 17.2.9 Franck-Condon Principle
- 17.2.10 Solved Problems Based on UV-Vis Spectroscopy
- 17.2.11 Applications of UV-Visible Spectroscopy
- 17.3 IR-Spectroscopy
- 17.4 Nuclear Magnetic Resonance Spectroscopy
- 17.4.1 Principle
- 17.4.2 Instrumentation
- 17.4.3 Chemical Shift
- 17.4.4 Spin-Spin Splitting, Spin-Spin Interaction, Spin–Spin Coupling or Fine Spectrum
- 17.4.5 Magnetic Resonance Imaging
- 17.4.6 High Resolution Proton Magnetic Resonance Spectroscopy
- 17.4.7 NMR Applications
- 17.4.8 Solved Problems Based on Proton NMR
- 17.5 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short Answer Questions
- Descriptive Questions
- 18. Thermal Analysis
- 19. Chromatography
- 20. Solid State and X-Ray Diffraction
- 21. Green Chemistry
- 21.1 Introduction
- 21.2 Twelve Principles of Green Chemistry
- 21.3 Importance of Green Synthesis
- 21.4 Greenhouse Concepts
- 21.5 Greenhouse Gases and Greenhouse Effect
- 21.6 Carbon Sequestration
- 21.7 Why Carbon Dioxide is a Major Problem
- 21.8 Review Questions
- Fill in the Blanks
- Multiple-choice Questions
- Short answer Questions
- Descriptive Questions
- Lab Manual
14
PROCEDURE FOR SYSTEMATIC QUALITATIVE ANALYSIS OF ORGANIC SUBSTANCES
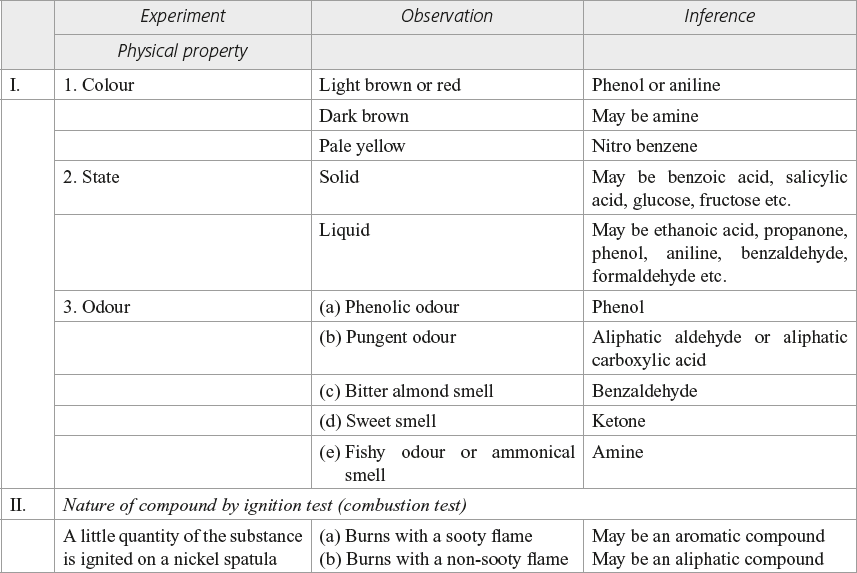





Report:
Name of the Compound:
Structural formula of the Compound:
Name of the prepared derivative:
SOME PRACTICAL EQUATIONS WITH DETAILED EXPLANATION
Solubility
Water
Phenol and formaldehyde can soluble in water due to formation hydrogen bonds.
Sodium hydroxide (NaOH)
Organic acids which are insoluble in water normally dissolved in NaOH solution due to the formation of respective conjugate base.

Dilute hydrochloric acid (HCl)
![]()
Sodium bicarbonate (NaHCO3)
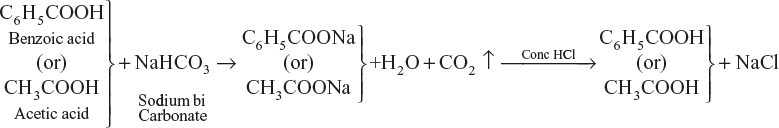
With dilute HCl, acetic acid does not regenerate.
Lassaigne’s test
Preparation of sodium fusion extract

- Na + C + N → NaCN
- 2Na + S → Na2S
- Na + X → NaX (Where X = Cl, Br, I)
N, S, Cl, Br are bonded with organic substances by covalent bonds; in the preparation of sodium fusion extract covalent bonds are converted into ionic bonds. Covalent bond is less reactive than ionic.
Test for nitrogen
![]()
In the presence of H2SO4. ferrous sulphate is converted into ferric sulphate.

Halogen
![]()
Sulphur
![]()
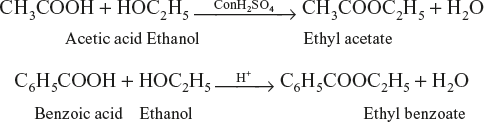
Esterification
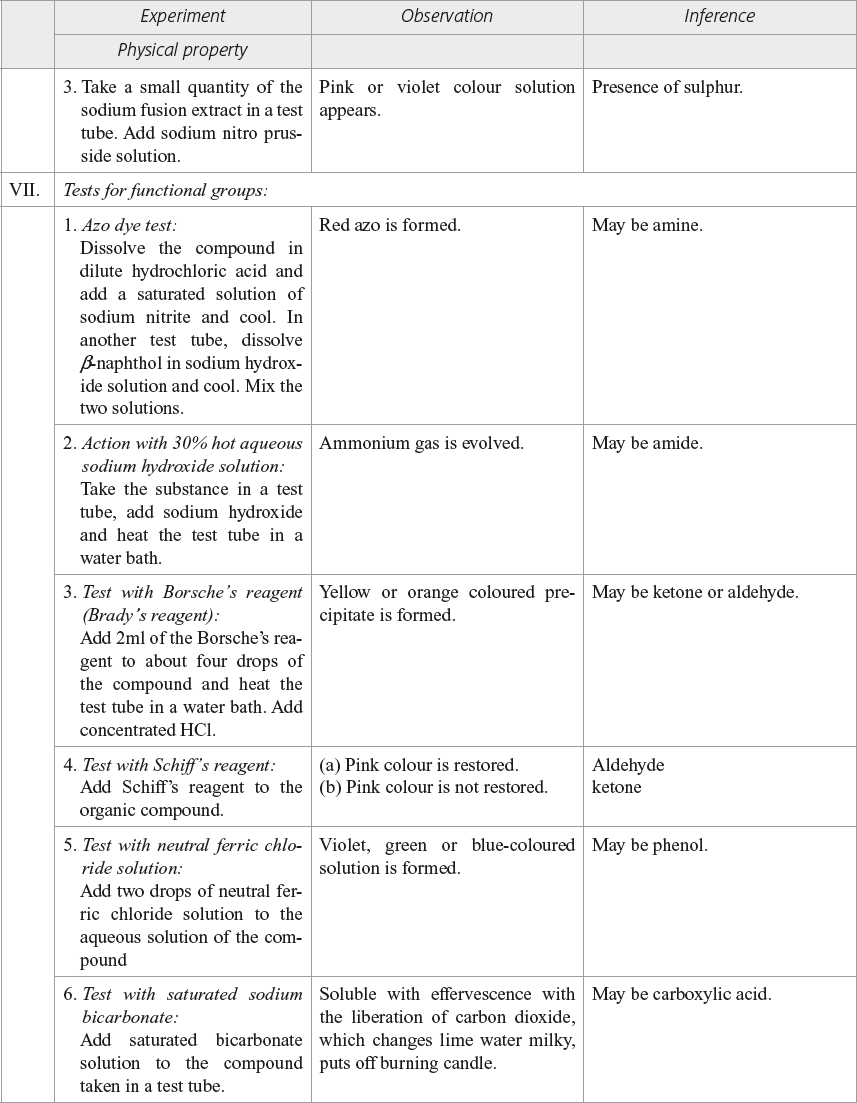
Borsche’s reagent
2, 4 Dinitro phenyl hydrazine (or) 2, 4 DNP

Tollen’s reagent
Ammonical solution of silver nitrate is known as Tollen’s reagent.
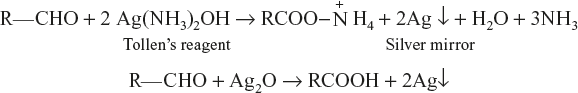
Fehling’s solution
Fehling’s solution is a blue coloured alkaline solution of cupric ion complexed with sodium potassium tartrate ions.

Benzaldehyde reduces only Tollen’s reagent.
Schiff’s reagent
A diluted solution of rosaniline hydrochloride,

Phenols azodye test

Bromination

Amines
Carbylamine reaction
![]()
Diazotisation

Amides

Derivatives
Amines
Bromo derivative

Acetyl derivative

Molisch’s test
Glucose CH2—OH—(CHOH)4—CHO

Reduction of Fehling solution

Reduction of Tollen’s reagent

2,4 dinitro phenyl hydrazine

Phenols derivatives
Benzoate derivative
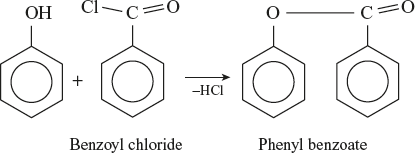
Reaction with benzyl chloride is known as schottan Baumann reaction.
Carboxylic acids
Anilide derivative

-
No Comment
