Appendix C. Strategies for Solving VLE Problems
In earlier chapters, we have discussed applications of VLE using simplified procedures such as bubble calculations using modified Raoult’s law. This appendix summarizes flow sheets for modified Raoult’s law and cubic equations of state.
• Section C.1 focuses on modified Raoult’s Law and offers a summary of flow sheets that quickly converge. Though you may be able to figure out other ways to converge calculations, it is often best to use strategies that have been well tested.
• Section C.2 discusses equations of state. Software permits solution of VLE using cubic equations without knowing the details of how the iterations are performed, and so a strategy is presented here also. There are many strategies throughout the literature, and the reader should be aware that other strategies are also successful.
• Section C.3 covers the gamma-phi method for nonideal gases with activity coefficients. These calculations require more sophistication, and are summarized online.
For each approach, there are five flowcharts presented—bubble P, bubble T, dew P, dew T, and isothermal flash. Specific routines may also be written for VLLE or other multiphase applications, which are not summarized here.
C.1. Modified Raoult’s Law Methods
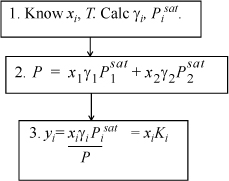
The equation that must be solved is: yiP = xiγiPisat
Bubble P
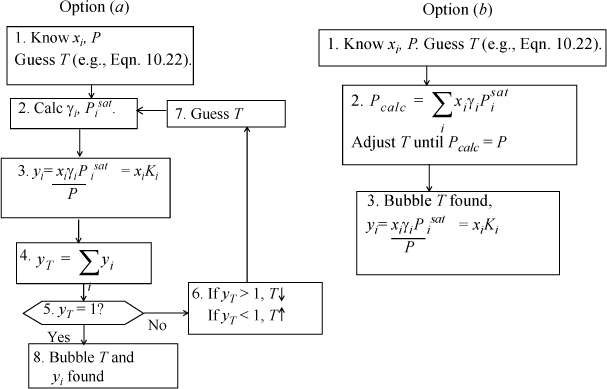
Bubble T
(Choose one flow sheet.)
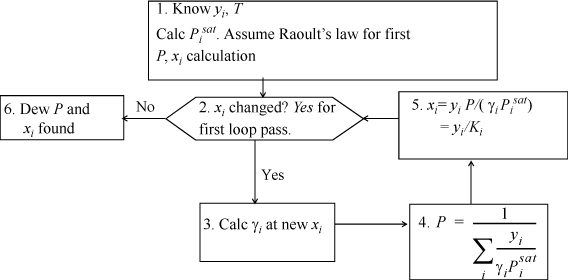
Dew P
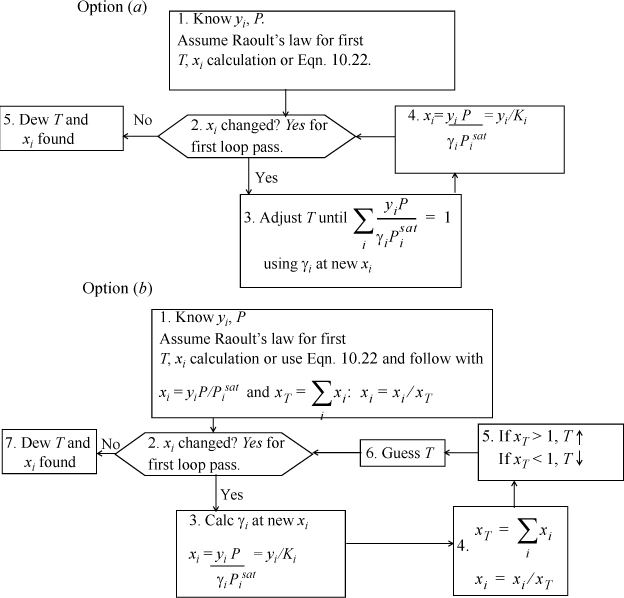
Dew T
(Choose one flow sheet.)
Isothermal Flash
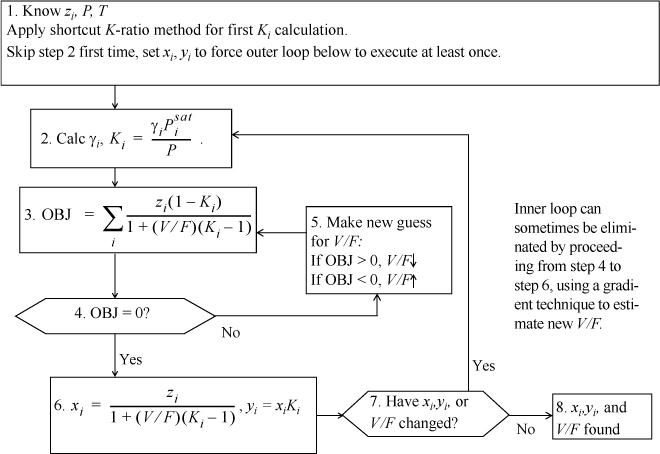
C.2. EOS Methods
The equation that must be solved is: ![]()
Bubble P
(The bubble pressure flow sheet is presented in Section 15.4)
Bubble T

Dew P
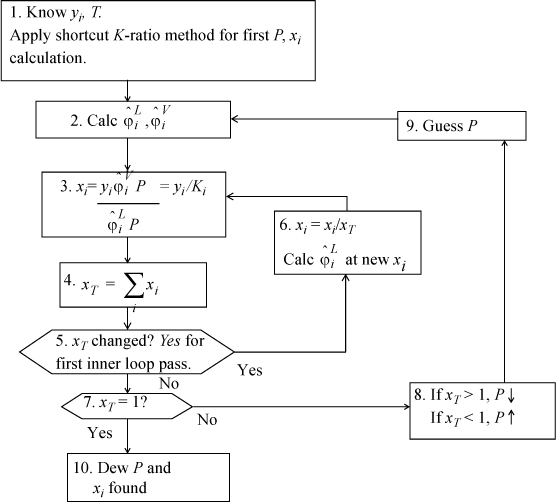
Dew T
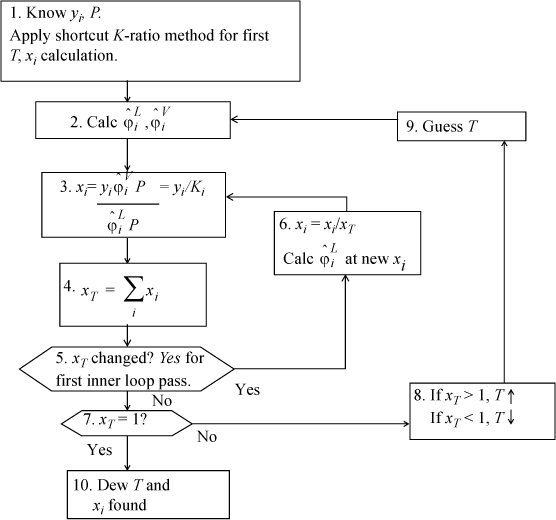
Isothermal Flash
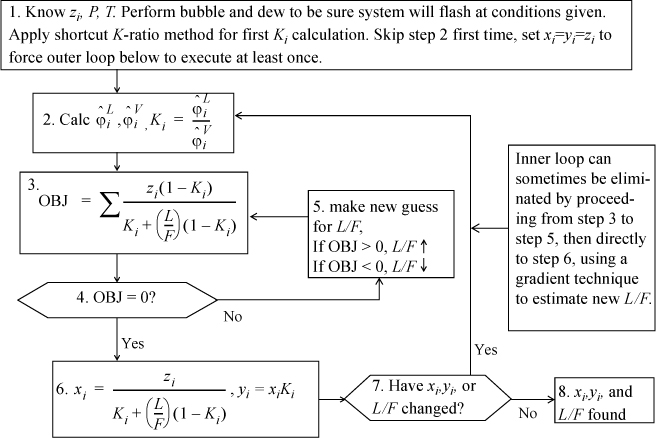
C.3. Activity Coefficient (Gamma-Phi) Methods
The equation that must be solved is: ![]() .
.
These flow sheets are available online as a supplement to Unit III.
