Appendix A: Viscosities and Other Properties of Gases and Liquids
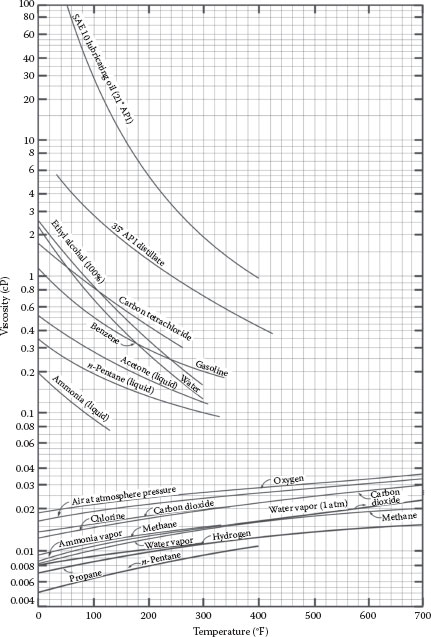
FIGURE A.1 Viscosities of various fluids at 1 atm pressure. 1 cP = 0.01 g/(cm s) = 6.72 × 10−4 lbm/(ft s) = 2.42 lbm/(ft h) = 2.09 × 10−5 lbf s/ft2. (Reproduced from Brown, G.G. et al., Unit Operations, Wiley, New York, 1951, p. 586. With permission.)
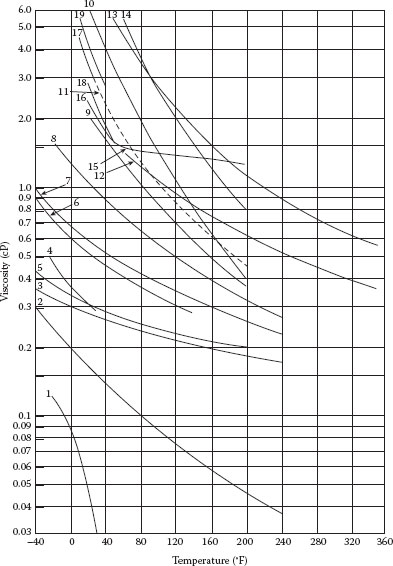
FIGURE A.2 Viscosity of various liquids: (1) Carbon dioxide, (2) ammonia, (3) methyl chloride, (4) sulfur dioxide, (5) Freon 12, (6) Freon 114, (7) Freon 11, (8) Freon 113, (9) ethyl alcohol, (10) isopropyl alcohol, (11) 20% sulfuric acid—20% H2SO4, (12) Dowtherm E, (13) Dowtherm A, (14) 20% sodium hydroxide—20% NaOH, (15) mercury, (16) 10% sodium chloride brine—10% NaCl, (17) 20% sodium chloride brine—20% NaCl, (18) 10% calcium chloride brine—10% CaCl2, and (19) 20% calcium chloride brine—20% CaCl2. (From Crane Technical Paper 4–10, Crane Co., Chicago, IL, 1991.)
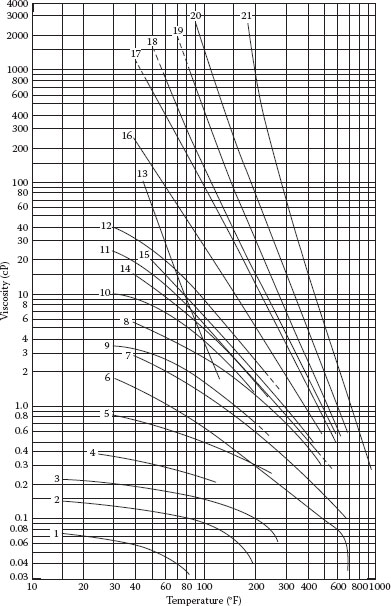
FIGURE A.3 Viscosity of water and liquid petroleum products: (1) Ethane (C2H6), (2) propane (C3H8), (3) butane (C4H10), (4) natural gasoline, (5) gasoline, (6) water, (7) kerosene, (8) distillate, (9) 48° API crude, (10) 40° API crude, (11) 35.6° API crude, (12) 32.6° API crude, (13) salt creek crude, (14) Fuel 3 (Max.), (15) Fuel 5 (Min.), (16) SAE 10 lube (100 V.I.), (17) SAE 30 lube (100 V.I.), (18) Fuel 5 (Min.) or Fuel 6 (Min.), (19) SAE 70 lube (100 V.I.), (20) Bunker C fuel (Max.) and M.C. Residuum, and (21) asphalt. (From Crane Company, Crane Technical Paper 410, Crane Co., Chicago, IL, 1991.)
TABLE A.1
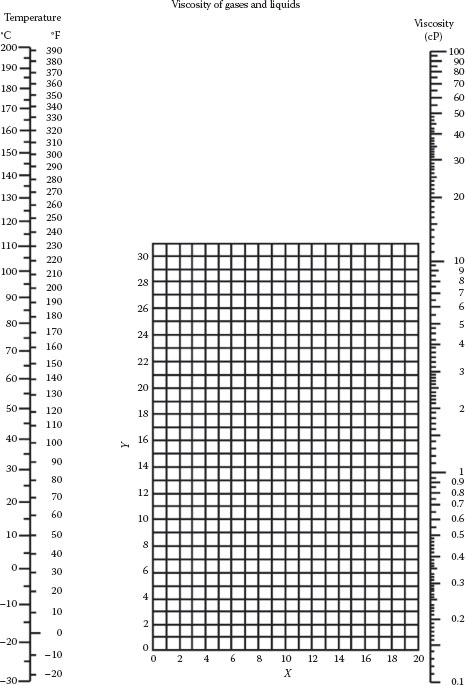
Viscosities of Liquids (Coordinates Apply to Figure A.4)
Liquid |
X |
Y |
Acetaldehyde |
15.2 |
4.8 |
Acetic acid, 100% |
12.1 |
14.2 |
Acetic acid, 70% |
9.5 |
17.0 |
Acetic anhydride |
12.7 |
12.8 |
Acetone, 100% |
14.5 |
7.2 |
Acetone, 35% |
7.9 |
15.0 |
Acetonitrile |
14.4 |
7.4 |
Acrylic acid |
12.3 |
13.9 |
Allyl alcohol |
10.2 |
14.3 |
Allyl bromide |
14.4 |
9.6 |
Allyl iodide |
14.0 |
11.7 |
Ammonia, 100% |
12.6 |
2.0 |
Ammonia, 26% |
10.1 |
13.9 |
Amyl acetate |
11.8 |
12.5 |
Amyl alcohol |
7.5 |
18.4 |
Aniline |
8.1 |
18.7 |
Anisole |
12.3 |
13.5 |
Arsenic trichloride |
13.9 |
14.5 |
Benzene |
12.5 |
10.9 |
Brine, CaCl(S)_2(S), 25% |
6.6 |
15.9 |
Brine, NaCl, 25% |
10.2 |
16.6 |
Bromine |
14.2 |
13.2 |
Bromotoluene |
20.0 |
15.9 |
Butyl acetate |
12.3 |
11.0 |
Butyl acrylate |
11.5 |
12.6 |
Butyl alcohol |
8.6 |
17.2 |
Butyric acid |
12.1 |
15.3 |
Carbon dioxide |
11.6 |
0.3 |
Carbon disulfide |
16.1 |
7.5 |
Carbon tetrachloride |
12.7 |
13.1 |
Chlorobenzene |
12.3 |
12.4 |
Chloroform |
14.4 |
10.2 |
Chlorosulfonic acid |
11.2 |
18.1 |
Chlorotoluene, ortho |
13.0 |
13.3 |
Chlorotoluene, meta |
13.3 |
12.5 |
Chlorotoluene, para |
13.3 |
12.5 |
Cresol, meta |
2.5 |
20.8 |
Cyclohexanol |
2.9 |
24.3 |
Cyclohexane |
9.8 |
12.9 |
Dibromomethane |
12.7 |
15.8 |
Dichloroethane |
13.2 |
12.2 |
Dichloromethane |
14.6 |
8.9 |
Diethyl ketone |
13.5 |
9.2 |
Diethyl oxalate |
11.0 |
16.4 |
Diethylene glycol |
5.0 |
24.7 |
Diphenyl |
12.0 |
18.3 |
Dipropyl ether |
13.2 |
8.6 |
Dipropyl oxalate |
10.3 |
17.7 |
Ethyl acetate |
13.7 |
9.1 |
Ethyl acrylate |
12.7 |
10.4 |
Ethyl alcohol, 100% |
10.5 |
13.8 |
Ethyl alcohol, 95% |
9.8 |
14.3 |
Ethyl alcohol, 40% |
6.5 |
16.6 |
Ethyl benzene |
13.2 |
11.5 |
Ethyl bromide |
14.5 |
8.1 |
2-Ethyl butyl acrylate |
11.2 |
14.0 |
Ethyl chloride |
14.8 |
6.0 |
Ethyl ether |
14.5 |
5.3 |
Ethyl formate |
14.2 |
8.4 |
2-Ethyl hexyl acrylate |
9.0 |
15.0 |
Ethyl iodide |
14.7 |
10.3 |
Ethyl propionate |
13.2 |
9.9 |
Ethyl propyl ether |
14.0 |
7.0 |
Ethyl sulfide |
13.8 |
8.9 |
Ethylene bromide |
11.9 |
15.7 |
Ethylene chloride |
12.7 |
12.2 |
Ethylene glycol |
6.0 |
23.6 |
Ethylidene chloride |
14.1 |
8.7 |
Fluorobenzene |
13.7 |
10.4 |
Formic acid |
10.7 |
15.8 |
Freon-11 |
14.4 |
9.0 |
Freon-12 |
16.8 |
5.6 |
Freon-21 |
15.7 |
7.5 |
Freon-22 |
17.2 |
4.7 |
Freon-113 |
12.5 |
11.4 |
Glycerol, 100% |
2.0 |
30.0 |
Glycerol, 50% |
6.9 |
19.6 |
Heptane |
14.1 |
8.4 |
Hexane |
14.7 |
7.0 |
Hydrochloric acid, 31.5% |
13.0 |
16.6 |
Iodobenzene |
12.8 |
15.9 |
Isobutyl alcohol |
7.1 |
18.0 |
Isobutyric acid |
12.2 |
14.4 |
Isopropyl alcohol |
8.2 |
16.0 |
Isopropyl bromide |
14.1 |
9.2 |
Isopropyl chloride |
13.9 |
7.1 |
Isopropyl iodide |
13.7 |
11.2 |
Kerosene |
10.2 |
16.9 |
Linseed oil, raw |
7.5 |
27.2 |
Mercury |
18.4 |
16.4 |
Methanol, 100% |
12.4 |
10.5 |
Methanol, 90% |
12.3 |
11.8 |
Methanol, 40% |
7.8 |
15.5 |
Methyl acetate |
14.2 |
8.2 |
Methyl acrylate |
13.0 |
9.5 |
Methyl r-butyrate |
12.3 |
9.7 |
Methyl n-butyrate |
13.2 |
10.3 |
Methyl chloride |
15.0 |
3.8 |
Methyl ethyl ketone |
13.9 |
8.6 |
Methyl formate |
14.2 |
7.5 |
Methyl iodide |
14.3 |
9.3 |
Methyl propionate |
13.5 |
9.0 |
Methyl propyl ketone |
14.3 |
9.5 |
Methyl sulfide |
15.3 |
6.4 |
Naphthalene |
7.9 |
18.1 |
Nitric acid |
12.8 |
13.8 |
Nitric acid, 60% |
10.8 |
17.0 |
Nitrobenzene |
10.6 |
16.2 |
Nitrogen dioxide |
12.9 |
8.6 |
Nitrotoluene |
11.0 |
17.0 |
Octane |
13.7 |
10.0 |
Octyl alcohol |
6.6 |
21.1 |
Pentachloroethane |
10.9 |
17.3 |
Pentane |
14.9 |
5.2 |
Phenol |
6.9 |
20.8 |
Phosphorus tribromide |
13.8 |
16.7 |
Phosphorus trichloride |
16.2 |
10.9 |
Propionic acid |
12.8 |
13.8 |
Propyl acetate |
13.1 |
10.3 |
Propyl alcohol |
9.1 |
16.5 |
Propyl bromide |
14.5 |
7.5 |
Propyl chloride |
14.4 |
7.5 |
Propyl formate |
13.1 |
9.7 |
Propyl iodide |
14.1 |
11.6 |
Sodium |
16.4 |
13.9 |
Sodium hydroxide, 50% |
3.2 |
25.8 |
Stannic chloride |
13.5 |
12.8 |
Succinonitrile |
10.1 |
20.8 |
Sulfur dioxide |
15.2 |
7.1 |
Sulfuric acid, 110% |
7.2 |
27.4 |
Sulfuric acid, 100% |
8.0 |
25.1 |
Sulfuric acid, 98% |
7.0 |
24.8 |
Sulfuric acid, 60% |
10.2 |
21.3 |
Sulfuryl chloride |
15.2 |
12.4 |
Tetrachloroethane |
11.9 |
15.7 |
Thiophene |
13.2 |
11.0 |
Titanium tetrachloride |
14.4 |
12.3 |
Toluene |
13.7 |
10.4 |
Trichloroethylene |
14.8 |
10.5 |
Triethylene glycol |
4.7 |
24.8 |
Turpentine |
11.5 |
14.9 |
Vinyl acetate |
14.0 |
8.8 |
Vinyl toluene |
13.4 |
12.0 |
Water |
10.2 |
13.0 |
Xylene, ortho |
13.5 |
12.1 |
Xylene, meta |
13.9 |
10.6 |
Xylene, para |
13.9 |
10.9 |
Source: Perry, R.H. and Green, D.W. (eds.), Perry’s Chemical Engineers’ Handbook, 7th edn., McGraw-Hill, New York, 1997. With permission.
FIGURE A.4 Nomograph for viscosities of liquids at 1 atm. See Table A.1 for coordinates. (To convert centipoise to pascal-seconds, multiply by 0.001.)
TABLE A.2
The Viscosity of Water at 0°C–100°C
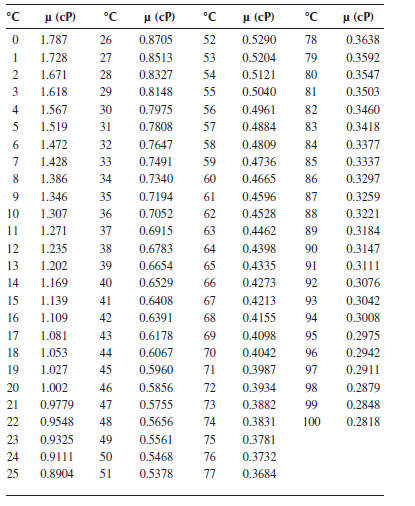
Table entries were calculated from the following empirical relationships from measurements in viscometers calibrated with water at 20°C (and 1 atm), modified to agree with the currently accepted value for the viscosity at 20°C of 1.002 cP:
(Hardy and Cottingham, 1949)
TABLE A.3
Physical Properties of Ordinary Water and Common Liquids (SI Units)
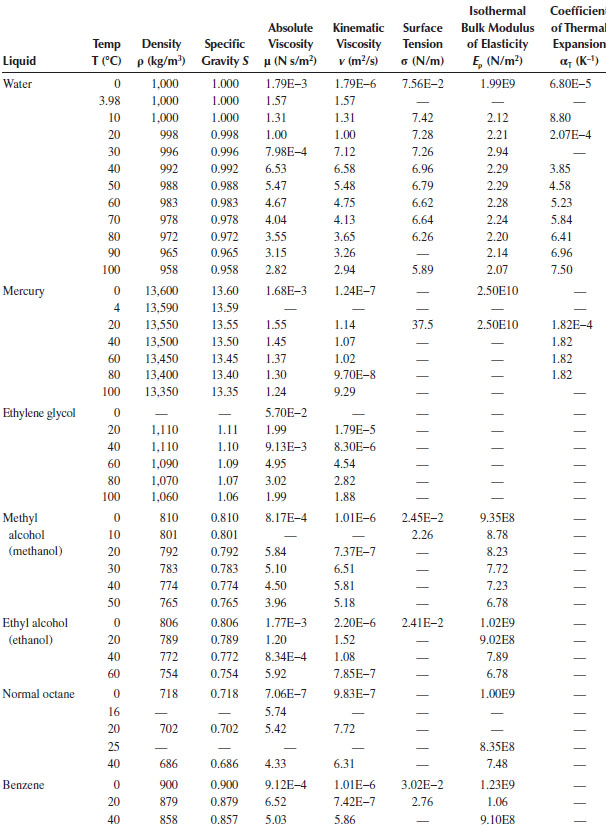
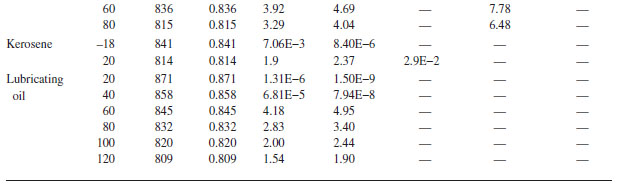
TABLE A.4
Physical Properties of Ordinary Water and Common Liquids (EE Unitsa)
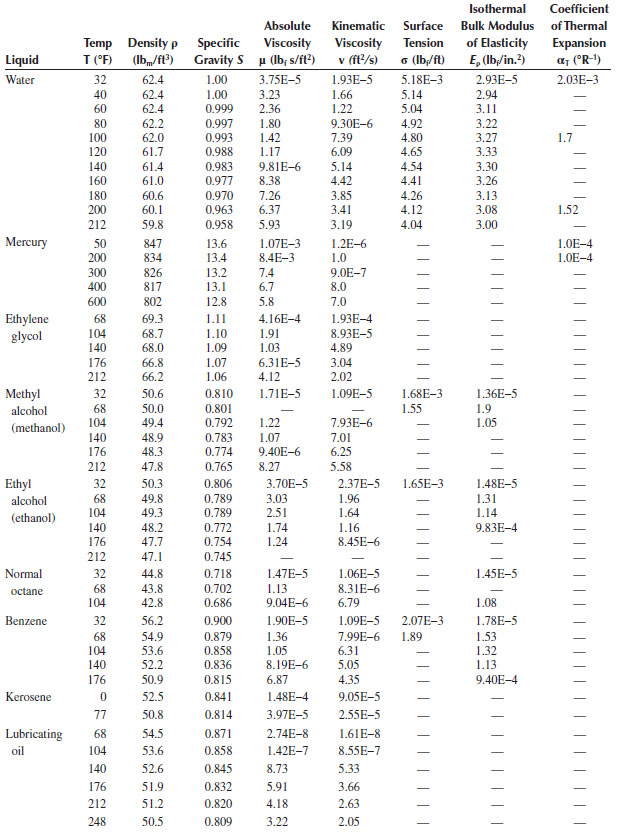
a EE, English engineering.
TABLE A.5
Physical Properties of SAE Oils and Lubricants
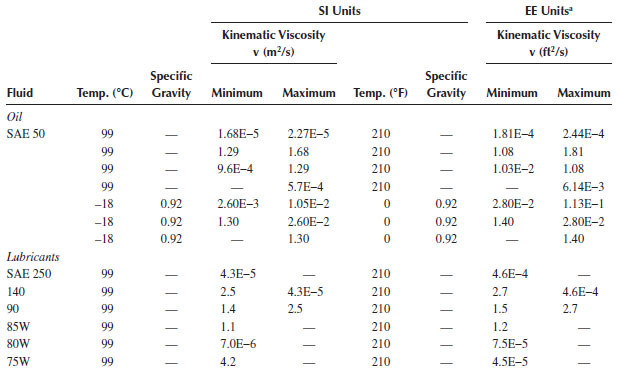
a EE, English engineering.
TABLE A.6
Viscosity of Steam and Watera
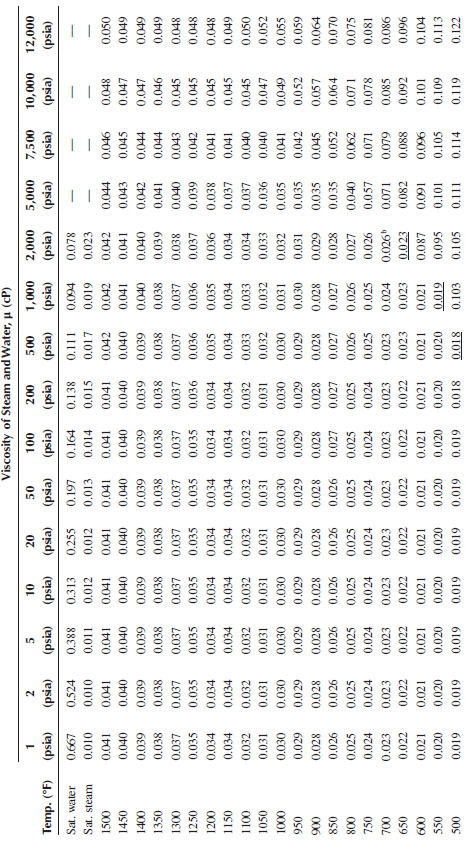

a Values directly below underscored viscosities are for water.
b Critical point.
TABLE A.7
Viscosities of Gasesa (Coordinates Apply to Figure A.5)
Gas |
X |
Y |
μ × 107 (P) |
Acetic acid |
7.0 |
14.6 |
825 (50°C) |
Acetone |
8.4 |
13.2 |
735 |
Acetylene |
9.3 |
15.5 |
1017 |
Air |
10.4 |
20.4 |
1812 |
Ammonia |
8.4 |
16.0 |
1000 |
Amylene (β) |
8.6 |
12.2 |
676 |
Argon |
9.7 |
22.6 |
2215 |
Arsine |
8.6 |
20.0 |
1575 |
Benzene |
8.7 |
13.2 |
746 |
Bromine |
8.8 |
19.4 |
1495 |
Butane (η) |
8.6 |
13.2 |
735 |
Butane (iso) |
8.6 |
13.2 |
744 |
Butyl acetate (iso) |
5.7 |
16.3 |
778 |
Butylene (α) |
8.4 |
13.5 |
761 |
Butylene (β) |
8.7 |
13.1 |
746 |
Butylene (iso) |
8.3 |
13.9 |
786 |
Butyl formate (iso) |
6.6 |
16.0 |
840 |
Cadmium |
7.8 |
22.5 |
5690 (500) |
Carbon dioxide |
8.9 |
19.1 |
1463 |
Carbon disulfide |
8.5 |
15.8 |
990 |
Carbon monoxide |
10.5 |
20.0 |
1749 |
Carbon oxysulfide |
8.2 |
17.9 |
1220 |
Carbon tetrachloride |
8.0 |
15.3 |
966 |
Chlorine |
8.8 |
18.3 |
1335 |
Chloroform |
8.8 |
15.7 |
1000 |
Cyanogen |
8.2 |
16.2 |
1002 |
Cyclohexane |
9.0 |
12.2 |
701 |
Cyclopropane |
8.3 |
14.7 |
870 |
Deuterium |
11.0 |
16.2 |
1240 |
Diethyl ether |
8.8 |
12.7 |
730 |
Dimethyl ether |
9.0 |
15.0 |
925 |
Diphenyl ether |
8.6 |
10.4 |
610 (50) |
Diphenyl methane |
8.0 |
10.3 |
605 (50) |
Ethane |
9.0 |
14.5 |
915 |
Ethanol |
8.2 |
14.5 |
835 |
Ethyl acetate |
8.4 |
13.4 |
743 |
Ethyl chloride |
8.5 |
15.6 |
987 |
Ethylene |
9.5 |
15.2 |
1010 |
Ethyl propionate |
12.0 |
12.4 |
890 |
Fluoride |
7.3 |
23.8 |
2250 |
Freon-11 |
8.6 |
16.2 |
1298 (93) |
Freon-12 |
9.0 |
17.4 |
1496 (93) |
Freon-14 |
9.5 |
20.4 |
1716 |
Freon-21 |
9.0 |
16.7 |
1389 (93) |
Freon-22 |
9.0 |
17.7 |
1554 (93) |
Freon-113 |
11.0 |
14.0 |
1166 (93) |
Freon-114 |
9.4 |
16.4 |
1364 (93) |
Helium |
11.3 |
20.8 |
1946 |
Heptane (n) |
9.6 |
10.6 |
618 (50) |
Hexane (n) |
8.4 |
12.0 |
644 |
Hydrogen |
11.3 |
12.4 |
880 |
Hydrogen–helium |
|||
10% H2, 90% He |
11.0 |
20.5 |
1780 (0) |
25% H2, 75% He |
11.0 |
19.4 |
1603 (0) |
40% H2, 60% He |
10.7 |
18.4 |
1431 (0) |
60% H2, 40% He |
10.8 |
16.7 |
1227 (0) |
81% H2, 19% He |
10.5 |
15.0 |
1016 (0) |
Hydrogen–sulfur dioxide |
8.7 |
18.1 |
1259 (17) |
10% H2, 90% SO2 |
8.7 |
18.1 |
1259 (17) |
20% H2, 80% SO2 |
8.6 |
18.2 |
1277 (17) |
50% H2, 50% SO2 |
8.9 |
18.3 |
1332 (17) |
80% H2, 20% SO2 |
9.7 |
17.7 |
1306 (17) |
Hydrogen bromide |
8.4 |
21.6 |
1843 |
Hydrogen chloride |
8.5 |
19.2 |
1425 |
Hydrogen cyanide |
7.1 |
14.5 |
737 |
Hydrogen iodide |
8.5 |
21.5 |
1830 |
Hydrogen sulfide |
8.4 |
18.0 |
1265 |
Iodine |
8.7 |
18.7 |
1730 (100) |
Krypton |
9.4 |
24.0 |
2480 |
Mercury |
7.4 |
24.9 |
4500 (200) |
Mercury bromide |
8.5 |
19.0 |
2253 |
Mercuric chloride |
7.7 |
18.7 |
2200 (200) |
Mercuric iodide |
8.4 |
18.0 |
2045 (200) |
Mesitylene |
9.5 |
10.2 |
660 (50) |
Methane |
9.5 |
15.8 |
1092 |
Methane (deuterated) |
9.5 |
17.6 |
1290 |
Methanol |
8.3 |
15.6 |
935 |
Methyl acetate |
8.4 |
14.0 |
870 (50) |
Methyl acetylene |
8.9 |
14.3 |
867 |
3-Methyl-1-butene |
8.0 |
13.3 |
716 |
Methyl butyrate (iso) |
6.6 |
15.8 |
824 |
Methyl bromide |
8.1 |
18.7 |
1327 |
Methyl bromide |
8.1 |
18.7 |
1327 |
Methyl chloride |
8.5 |
16.5 |
1062 |
3-Methylene-1-butene |
8.0 |
13.3 |
716 |
Methylene chloride |
8.5 |
15.8 |
989 |
Methyl formate |
5.1 |
18.0 |
923 |
Neon |
11.1 |
25.8 |
3113 |
Nitric oxide |
10.4 |
20.8 |
1899 |
Nitrogen |
10.6 |
20.0 |
1766 |
Nitrous oxide |
9.0 |
19.0 |
1460 |
Nonane (n) |
9.2 |
8.9 |
554 (50) |
Octane (n) |
8.8 |
9.8 |
586 (50) |
Oxygen |
10.2 |
21.6 |
2026 |
Pentene (n) |
8.5 |
12.3 |
668 |
Pentane (iso) |
8.9 |
12.1 |
685 |
Phosphene |
8.8 |
17.0 |
1150 |
Propane |
8.9 |
13.5 |
800 |
Propanol (n) |
8.4 |
13.5 |
770 |
Propanol (iso) |
8.4 |
13.6 |
774 |
Propyl acetate |
8.0 |
14.3 |
797 |
Propylene |
8.5 |
14.4 |
840 |
Pyridine |
8.6 |
13.3 |
830 (50) |
Silane |
9.0 |
16.8 |
1148 |
Stannic chloride |
9.1 |
16.0 |
1330 (100) |
Stannic bromide |
9.0 |
16.7 |
142 (100) |
Sulfur dioxide |
8.4 |
18.2 |
1250 |
Thiazole |
10.0 |
14.4 |
958 |
Thiophene |
8.3 |
14.2 |
901 (50) |
Toluene |
8.6 |
12.5 |
686 |
2,2,3-Trimethylbutane |
10.0 |
10.4 |
691 (50) |
Trimethylethane |
8.0 |
13.0 |
686 |
Water |
8.0 |
16.0 |
1250 (100) |
Xenon |
9.3 |
23.0 |
2255 |
Zinc |
8.0 |
22.0 |
5250 (500) |
Source: Perry, R.H. and Green, D.W. (eds.), Perry’s Chemical Engineers’ Handbook, 7th edn., McGraw-Hill, New York, 1997. With permission.
a Viscosity at 20°C unless otherwise indicated.
FIGURE A.5 Nomograph for determining the absolute viscosity of a gas near ambient pressure and the relative viscosity of a gas compared with air. (Coordinates from Table A.7.) To convert from poise to pascal-seconds, multiply by 0.1. (From Beerman, Meas. Control., June 1982, 154–157.)
A.1 VISCOSITIES OF GASES AND LIQUIDS
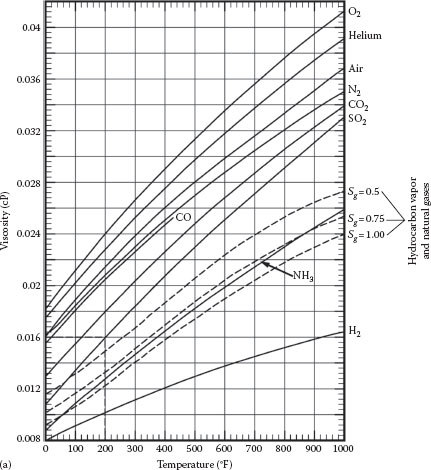
The curves for hydrocarbon vapors and natural gases in the chart at the upper right are taken from Maxwell, and the curves for all other gases (except helium) in the chart are based upon Sutherland’s formula, as follows:
where
μ is the viscosity in cP at temperature T
μ0 is the viscosity in cP at temperature T0
T is the absolute temperature, in °R (460 + °F), for which viscosity is desired
T0 is the absolute temperature, in °R, for which viscosity is known
C is Sutherland’s constant
FIGURE A.6 Viscosity of (a) gases.
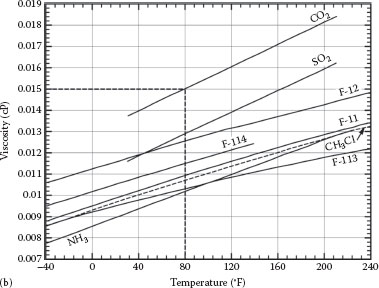
FIGURE A.6 Viscosity of (a) gases. (b) refrigerant vapors. (From Crane Technical Paper 410, Crane Co., Chicago, IL, 1991.)
Note: The variation of viscosity with pressure is small for most gases. For gases given on this page, the correction of viscosity for pressure is less than 10% for pressures up to 500 psi.
Fluid |
Approximate Values of C |
O2 |
127 |
Air |
120 |
N2 |
111 |
CO2 |
240 |
CO |
118 |
SO2 |
416 |
NH3 |
370 |
H2 |
72 |
Example (Figure A.6a): The viscosity of sulfur dioxide gas (SO2) at 200°F is 0.016 cP.
Example (Figure A.6b): The viscosity of carbon dioxide gas (CO2) at about 80°F is 0.015 cP.
REFERENCES
Brown, G.G. et al., Unit Operations, Wiley, New York, 1951, p. 586.
Crane Company, Crane Technical Paper 410, Crane Co., Chicago, IL, 1991.
Hardy, R.C. and R.L. Cottingham, Viscosity of Water at T −20 to 150°C, J. Res. NBS, 42, 573, 1949.
Perry, R.H. and Green, D.W. (eds.), Perry’s Chemical Engineers’ Handbook, 7th edn., McGraw-Hill, New York, 1997.