Chapter 4
Surface Tension Determination
INTRODUCTION
Surface tension is a phenomenon in which the surface of the liquid is in contact with the gas. Surface tension is defined as the force acting at right angles to a line of unit length present in the surface. This is denoted by ‘γ’.
where W = work; F = force; d = distance.
Therefore,
The SI units for the surface tension are newton/meter or dyne/cm.
At a liquid surface, the molecule is not completely surrounded by other molecules.
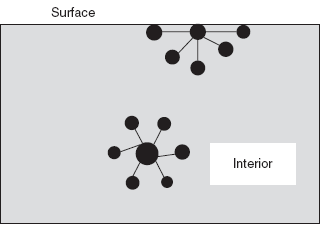
Surface tension phenomenon
The surface tension of a liquid is the energy required to increase the surface area which increases the stability of the solution and increases the solubility rate.
Examples of surface tension are drops of water, insects walking on water and paper clips floating on water.
An important measure for the characterization of surfactants which reduces surface tension is the critical micelle concentration (CMC). These surfactants contain both hydrophobic and hydrophilic groups attached to the surface. CMC is defined as the concentration of the surfactant at which the micelle formation starts.
The main cause for the surface tension is van der Waal's force which draws the molecules together.
FACTORS AFFECTING SURFACE TENSION
- The stronger the intermolecular force, the higher the surface tension
- Increased temperature decreases surface tension
- Surfactants present in the solution reduce surface tension
- Increased molecular motion increases surface tension
METHODS FOR DETERMINING SURFACE TENSION
There are different methods for the determination of surface tension, as follows:
Du Nouy Ring Method
This method was first proposed by the Pierre Lecomte du Nouy in 1883. The basic principle involved in this method is lifting a ring from the surface of a liquid. Then the slow lifting of the ring which is madeup of platinum from the liquid surface is observed. The force required to raise the ring from the liquid surface is measured.
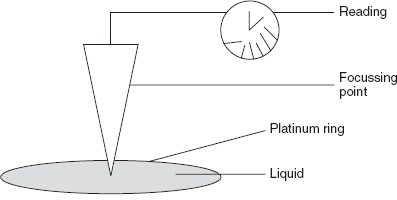
Du Nouy ring apparatus
Drop Shape Method
Surface tension is determined by fitting of the shape of the drop to the Young–Laplace equation:
where ΔP = pressure difference; γ = surface tension; R1 and R2 = surfaces radii of curvature.
Wilhelmy Plate Method
This method was first proposed by Ludwig Wilhelmy. A Wilhelmy plate is a thin plate used to measure equilibrium surface tension between the air–liquid or liquid–liquid interfaces.
where l = wetted perimeter of plate; θ = contact angle between the interfaces of liquid and plate; F = force.
Stalagmometric Method
The stalagmometer consists of the glass capillary which is having the marks. The stalagmometer is filled with the sample solution and measures the weight of the drops and the number of drops. Based on this method, there are two other types:
- Drop weight method
- Drop number method
Drop weight method
The stalagmometer is filled with water and then allowed to run from Mark A to Mark B. The method follows the stalagmometer method and is rinsed with water. The weigh bottle is taken and the weight of the empty bottle is noted as W1. Then the collected water is taken into the weigh bottle and the weight is noted as W2. Then the stalagmometer is filled with the sample solution and is allowed to run from Mark A to Mark B. Then collect in the weigh bottle and take the weight as W3. Then the surface tension is determined by the following equation:
Drop count method
The procedure is the same as for the drop count method. However, the number of drops is counted instead of the weight. Then the number of drops is counted from the liquid flown from Mark A to Mark B. Then the surface tension is determined from the following equation:
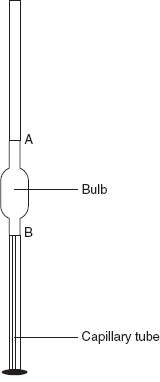
Stalagmometer apparatus
APPLICATIONS
- Used in the determination of different surfactant critical micelle concentrations
- Used in the removal of dust particles
- Used in the soap and detergent manufacturing industry
- Used in the determination of surface tensions of the liquid–liquid or air–liquid interfaces
REVIEW QUESTIONS
- Define surface tension.
- What are the factors affecting surface tension?
- What is the principle involved in the du Nouy tensiometer?
- Write the equations for the drop count and drop weight methods.
- What is the principle involved in the Wilhelmy plate method?
- What are the applications of surface tension determinations?
