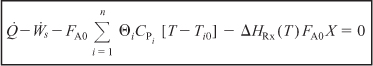11.3.2 Dissecting the Enthalpies
We are neglecting any enthalpy changes resulting from mixing so that the partial molal enthalpies are equal to the molal enthalpies of the pure components. The molal enthalpy of species i at a particular temperature and pressure, Hi, is usually expressed in terms of an enthalpy of formation of species i at some reference temperature TR, ![]() , plus the change in enthalpy ΔHQi, that results when the temperature is raised from the reference temperature, TR, to some temperature T:
, plus the change in enthalpy ΔHQi, that results when the temperature is raised from the reference temperature, TR, to some temperature T:
![]()
The reference temperature at which ![]() is given is usually 25°C. For any substance i that is being heated from T1 to T2 in the absence of phase change,
is given is usually 25°C. For any substance i that is being heated from T1 to T2 in the absence of phase change,
![]()
Typical units of the heat capacity, CPi, are
![]()
A large number of chemical reactions carried out in industry do not involve phase change. Consequently, we shall further refine our energy balance to apply to single-phase chemical reactions. Under these conditions, the enthalpy of species i at temperature T is related to the enthalpy of formation at the reference temperature TR by
![]()
If phase changes do take place in going from the temperature for which the enthalpy of formation is given and the reaction temperature T, Equation (11-17) must be used instead of Equation (11-19).
The heat capacity at temperature T is frequently expressed as a quadratic function of temperature, that is,
![]()
However, while the text will consider only constant heat capacities, the PRS R11.3 on the DVD-ROM has examples with variable heat capacities.
To calculate the change in enthalpy (Hi – Hi0) when the reacting fluid is heated without phase change from its entrance temperature, Ti0, to a temperature T, we integrate Equation (11-19) for constant CPi to write

Substituting for Hi and Hi0 in Equation (11-16) yields